 Back
BackEnzymes and Biological Catalysis: Structure, Function, and Factors Affecting Activity
Study Guide - Smart Notes

Enzymes & Biological Rates
Introduction to Enzymes
Enzymes are specialized biological catalysts that dramatically increase the speed of chemical reactions essential for life. Unlike other proteins, enzymes are not consumed or permanently altered during the reactions they facilitate, allowing them to be reused repeatedly.
Biological Catalysts: Enzymes accelerate reactions by lowering the activation energy required for the process.
Protein-Based: Most enzymes are globular proteins with complex three-dimensional structures, which are crucial for their specificity and function.
Specificity: Each enzyme is tailored to catalyze a particular reaction or type of reaction, determined by its unique structure.
Structure and Function of Enzymes
The Active Site and Substrate Binding
The active site is a uniquely shaped region on the enzyme where the substrate—the molecule upon which the enzyme acts—binds. The precise fit between the enzyme and substrate ensures high specificity, often described as a "lock and key" or "induced fit" model.
Active Site: The region where substrate binding and catalysis occur.
Substrate: The specific molecule that the enzyme acts upon.
Enzyme-Substrate Complex: The temporary association formed when the substrate binds to the enzyme's active site.
How Enzymes Work: Lowering Activation Energy
Enzymes increase reaction rates by lowering the activation energy barrier, making it easier for reactants to be converted into products. This allows reactions that would otherwise take millions of years to occur in milliseconds, supporting the dynamic processes of life.
Activation Energy: The minimum energy required to initiate a chemical reaction.
Effect of Enzymes: Enzymes provide an alternative reaction pathway with a lower activation energy.
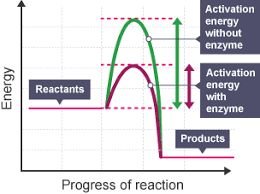
Additional info: The energy diagram illustrates how enzymes lower the activation energy, making reactions proceed faster without altering the overall energy change of the reaction.
Factors Affecting Enzyme Activity
Temperature Effects
Each enzyme has an optimal temperature at which its activity is highest. For human enzymes, this is typically around 37°C. Temperatures above this optimum can disrupt the hydrogen bonds maintaining the enzyme's structure, leading to denaturation and loss of function.
Optimum Temperature: The temperature at which enzyme activity is maximal.
Denaturation: The process by which proteins lose their structure and function due to the breaking of non-covalent bonds (e.g., hydrogen bonds) at high temperatures.
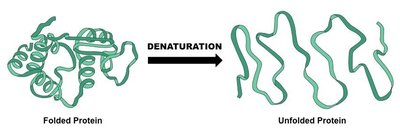
Example: Cooking an egg causes the proteins in the egg white to denature, changing from a transparent, soluble form to an opaque, solid form as the proteins unfold and aggregate.
pH Effects
Enzymes are also sensitive to pH, with each enzyme having an optimal pH range. Deviations from this range can alter the enzyme's structure and reduce its activity.
Enzyme Name | Found In | Optimal pH Range |
|---|---|---|
Pepsin | Stomach (acidic) | 1.5 – 2.0 |
Salivary Amylase | Mouth (neutral) | 6.7 – 7.0 |
Trypsin | Small Intestine (basic) | 7.8 – 8.5 |
Arginase | Liver (very basic) | 9.5 – 10.0 |
Additional info: The optimal pH reflects the environment in which the enzyme naturally functions.
Concentration Effects
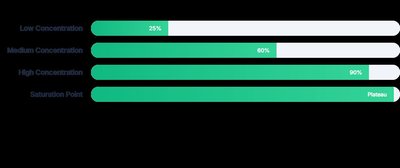
The rate of an enzyme-catalyzed reaction depends on both enzyme and substrate concentrations. Increasing enzyme concentration increases the reaction rate linearly, provided there is excess substrate. However, increasing substrate concentration only increases the rate up to a saturation point, beyond which all active sites are occupied and the rate plateaus.
Enzyme Concentration: More enzyme molecules provide more active sites, increasing the reaction rate if substrate is abundant.
Substrate Concentration: The reaction rate increases with substrate concentration until all enzyme active sites are saturated.
Saturation Point: The maximum rate of reaction, where increasing substrate concentration further has no effect.
Examples of Enzymes and Their Roles
Common Enzymes in Biological Systems
Amylase: Breaks down starch into simple sugars; found in saliva and pancreas.
Lipase: Breaks down lipids (fats) into fatty acids and glycerol; produced in the pancreas and intestine.
Catalase: Decomposes hydrogen peroxide into water and oxygen, protecting cells from oxidative damage.
Lactase: Breaks down lactose (milk sugar) into glucose and galactose.
DNA Polymerase: Synthesizes new DNA strands by adding nucleotides, essential for DNA replication and cell division.
The Catalytic Cycle of Enzymes
Steps in Enzyme Action
The catalytic cycle describes the sequence of events during enzyme-mediated reactions:
Binding: The substrate enters the enzyme's active site.
Enzyme-Substrate Complex: A temporary complex forms, positioning the substrate for reaction.
Catalysis: The enzyme facilitates bond breaking/forming, converting substrate to product.
Release: The product is released, and the enzyme is free to catalyze another reaction.
Summary Table: Factors Affecting Enzyme Activity
Factor | Effect on Enzyme Activity |
|---|---|
Temperature | Increases activity up to optimum; high temperatures cause denaturation |
pH | Each enzyme has an optimal pH; deviations reduce activity |
Enzyme Concentration | Increases rate if substrate is in excess |
Substrate Concentration | Increases rate up to saturation point |
Additional info: Enzyme kinetics and regulation are foundational concepts in both chemistry and biology, illustrating the interplay between structure, function, and environmental conditions.
