 Back
BackFactors Affecting the Rate of Chemical Reactions and Rate Curves
Study Guide - Smart Notes

Rate Curves in Chemical Reactions
Understanding Rate Curves
Rate curves are graphical representations that show how the progress of a chemical reaction changes over time. These curves can be used to monitor either the formation of products or the consumption of reactants, and their shape is influenced by various experimental conditions.
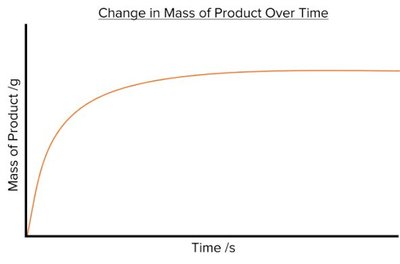
Product Formation: The mass or volume of product increases over time and eventually levels off when the reaction is complete.
Reactant Consumption: The amount of reactant decreases as the reaction proceeds, reaching zero when the limiting reagent is used up.
Key Features: Initial rate (steepness at the start), time to completion, and total amount of product formed.
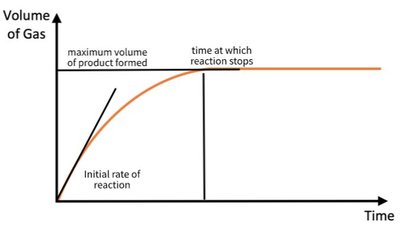
Limiting and Excess Reagents
Definitions and Effects
In a chemical reaction, the limiting reagent is the reactant that is completely consumed first, thus determining when the reaction stops and how much product is formed. The reactant in excess is the substance that remains after the reaction has stopped.
Limiting Reagent: Limits the amount of product; reaction stops when it is used up.
Reactant in Excess: Some remains unreacted; does not determine when the reaction stops.
Application: Identifying the limiting reagent is essential for calculating theoretical yield.

Factors Affecting Reaction Rate and Rate Curves
Concentration of a Reactant
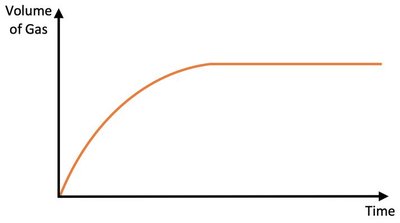
Increasing the concentration of a reactant generally increases the initial rate of reaction and can affect the time taken for the reaction to complete. If the concentration of the limiting reactant is increased, more product may be formed.
Effect: Higher concentration increases collision frequency between particles.
Result: Limiting reactant is used up faster; reaction completes sooner.
Graphical Representation: Steeper initial slope on rate curve.
Volume of a Reactant
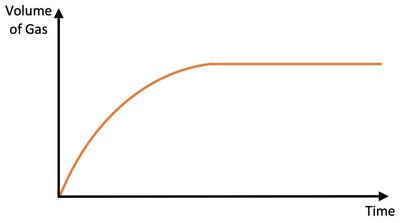
Increasing the volume of a reactant does not affect the initial rate but may increase the total amount of product formed if the volume increase involves the limiting reactant.
Effect: No change in collision frequency; initial rate unchanged.
Result: More product if limiting reactant volume is increased.
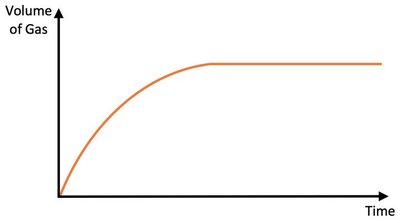
Temperature
Raising the temperature increases both the frequency of collisions and the proportion of particles with sufficient energy to react (activation energy, Ea). This increases the initial rate and shortens the time to completion, but does not affect the total amount of product formed.
Effect: More frequent and energetic collisions.
Result: Faster use of limiting reactant; reaction finishes sooner.
Equation: Arrhenius equation:
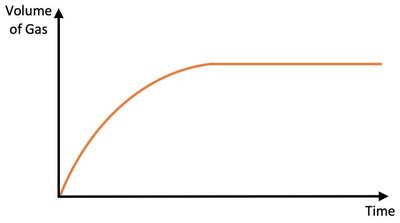
Surface Area of a Reactant
Increasing the surface area of a solid reactant (e.g., by using a powder instead of a lump) increases the initial rate of reaction by exposing more particles to collisions, but does not change the total amount of product formed.
Effect: More surface for collisions; higher initial rate.
Result: Limiting reactant is used up faster.
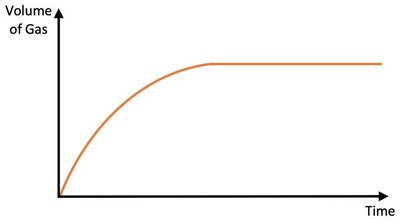
Mass of a Solid Reactant
Increasing the mass of a solid reactant can increase both the initial rate (due to increased surface area) and the total amount of product formed, especially if the solid is the limiting reactant.
Effect: More reactant particles and greater surface area.
Result: If solid is limiting, more product is formed; if not, reaction is faster but yield unchanged.
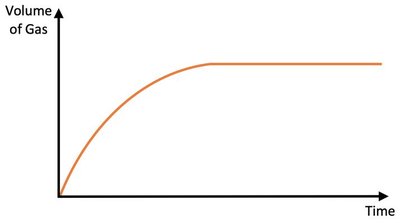
Adding a Catalyst
A catalyst increases the rate of a chemical reaction by lowering the activation energy required for the reaction to proceed. It does not affect the total amount of product formed or the position of equilibrium.
Effect: More particles have enough energy to react; faster reaction.
Result: Limiting reagent is used up faster; reaction completes sooner.
Equation: Catalysts lower Ea in the Arrhenius equation.
Summary Table: Effects of Different Factors on Reaction Rate
Factor | Initial Rate | Total Product Formed | Time to Completion |
|---|---|---|---|
Concentration (limiting reactant) | Increases | May increase | Decreases |
Volume (limiting reactant) | No change | Increases | May increase |
Temperature | Increases | No change | Decreases |
Surface Area | Increases | No change | Decreases |
Mass of Solid (if limiting) | Increases | Increases | May decrease |
Catalyst | Increases | No change | Decreases |
