 Back
BackGas Laws: Relationships Among Pressure, Volume, and Temperature
Study Guide - Smart Notes

The Gas Laws
Overview
The gas laws describe the relationships among the pressure, volume, and temperature of a gas. These laws are fundamental in understanding the behavior of gases and are essential for solving problems in chemistry involving gases.
Boyle’s Law: Relates pressure and volume at constant temperature.
Charles’s Law: Relates volume and temperature at constant pressure.
Gay-Lussac’s Law: Relates pressure and temperature at constant volume.
Combined Gas Law: Relates pressure, volume, and temperature for a fixed amount of gas.
Boyle’s Law
Pressure and Volume Relationship
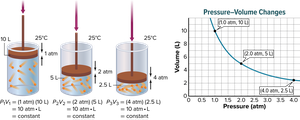
Boyle’s law states that the volume of a fixed amount of gas held at a constant temperature varies inversely with the pressure. This means that as pressure increases, volume decreases, and vice versa, provided temperature remains constant.
Mathematical Expression:
Inverse Relationship: If pressure doubles, volume halves.
Units: Pressure (atm, kPa), Volume (L, mL)
Example: A diver blows a 0.75-L air bubble 10 m under water. As it rises to the surface, the pressure goes from 2.25 atm to 1.03 atm. The volume at the surface can be calculated using Boyle’s law.
Charles’s Law
Temperature and Volume Relationship
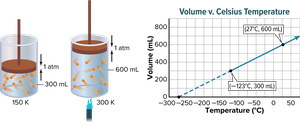
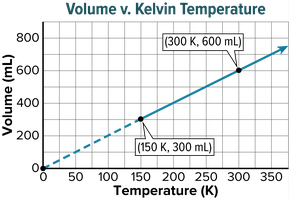
Charles’s law states that the volume of a given amount of gas is directly proportional to its Kelvin temperature at constant pressure. As temperature increases, so does the volume.
Mathematical Expression:
Direct Relationship: Doubling temperature (in Kelvin) doubles volume.
Absolute Zero: The lowest possible theoretical temperature, 0 K, where volume would be zero.
Example: A helium balloon in a closed car occupies a volume of 2.32 L at 40.0°C. If the temperature rises to 75.0°C, the new volume can be found using Charles’s law after converting temperatures to Kelvin.
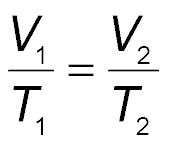
Gay-Lussac’s Law
Temperature and Pressure Relationship
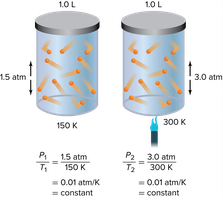
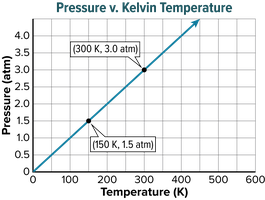
Gay-Lussac’s law states that the pressure of a fixed amount of gas varies directly with the Kelvin temperature when the volume remains constant. As temperature increases, pressure increases.
Mathematical Expression:
Direct Relationship: If temperature decreases, pressure decreases.
Example: The pressure of oxygen gas inside a canister is 5.00 atm at 25.0°C. If the temperature falls to -10.0°C, the new pressure can be calculated using Gay-Lussac’s law after converting temperatures to Kelvin.
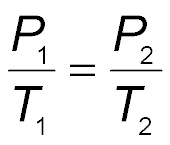
The Combined Gas Law
Relationship Among Pressure, Volume, and Temperature
The combined gas law expresses the relationship among pressure, volume, and temperature for a fixed amount of gas. It is useful when more than one variable changes.
Mathematical Expression:
Application: Used when pressure, volume, and temperature all change.
Example: A gas at 110 kPa and 30.0°C fills a container with a volume of 2.00 L. If the temperature rises to 80.0°C and the pressure increases to 440 kPa, the new volume can be calculated using the combined gas law.
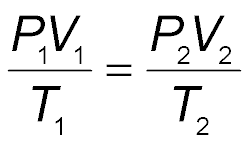
Key Terms and Concepts
Scientific Law: Describes a relationship in nature supported by many experiments.
Absolute Zero: The lowest possible theoretical temperature (0 K), where gas volume is zero.
Significant Figures: Answers to gas law problems should be expressed with the correct number of significant figures based on the data provided.
Quiz Review
Sample Questions
Boyle’s Law: What happens to the volume of a fixed amount of gas at constant temperature when the pressure doubles? Answer: The volume decreases by half.
Absolute Zero: Which is NOT true about absolute zero? Answer: It is zero on the Celsius scale.
Charles’s Law: Which law states that the volume of a given amount of gas is directly proportional to its Kelvin temperature at constant pressure? Answer: Charles’s law.
Gay-Lussac’s Law: Which variables remain constant when applying the formula for Gay-Lussac’s law? Answer: Amount of gas and volume.
