 Back
BackGases, Liquids, Solids, and Intermolecular Forces: Study Notes
Study Guide - Smart Notes

Gases, Liquids, Solids, and Intermolecular Forces
States of Matter
The three primary states of matter—solid, liquid, and gas—differ in the arrangement and movement of their particles. Understanding these differences is fundamental to chemistry.
Solids: Particles are closely packed in fixed positions and can only vibrate.
Liquids: Particles are close together but can move past one another, allowing liquids to flow.
Gases: Particles are far apart and move rapidly and randomly, filling any available space.

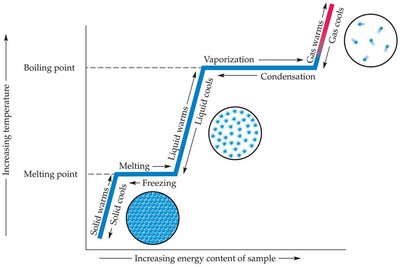
Phase Changes and Energy
Phase changes occur when matter transitions between solid, liquid, and gas states, typically due to changes in temperature or pressure. Each transition involves energy changes and specific terminology.
Melting point: The temperature at which a solid becomes a liquid.
Vaporization: The process by which a liquid becomes a gas.
Boiling point: The temperature at which a liquid's particles escape to become a gas.
Condensation: The process by which a gas becomes a liquid.
Freezing: The process by which a liquid becomes a solid (at the freezing/melting point).
Sublimation: The direct transition from solid to gas.
Intermolecular Forces
Types of Intermolecular Forces
Intermolecular forces are the attractions between molecules that determine the physical properties of substances, such as boiling and melting points.
Ionic bonds: The strongest forces, holding ions together in solids.
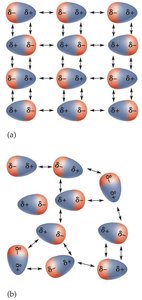
Dipole forces (Dipole-dipole interactions): Occur between polar molecules with permanent dipoles; oppositely charged ends attract each other.
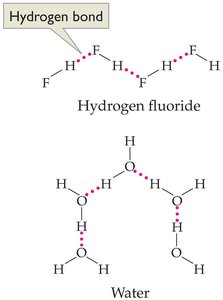
Hydrogen bonds: A special, strong type of dipole force occurring when hydrogen is bonded to highly electronegative atoms (N, O, or F). Responsible for many unique properties of water.
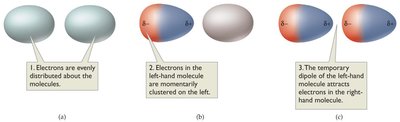
Dispersion forces (London dispersion forces): Weak attractions present in all molecules, especially significant in nonpolar molecules. Caused by temporary, induced dipoles that increase with molecular size.
Applications: Solutions and Solubility
Solutions are homogeneous mixtures of two or more substances. The ability of substances to form solutions depends on the similarity of their intermolecular forces.
Solute: The substance dispersed in a solution.
Solvent: The substance present in greater quantity, doing the dissolving.
"Like dissolves like": Solutions form most readily when solute and solvent have similar intermolecular forces (e.g., polar with polar, nonpolar with nonpolar).

The Gas Laws
Kinetic–Molecular Theory of Gases
The kinetic–molecular theory explains the behavior of gases based on the motion of their particles.
Gas particles are in rapid, constant motion.
Gas particles are tiny compared to the distances between them.
There is little attraction between gas particles.
Collisions between gas molecules are perfectly elastic (no energy lost).
Temperature measures the average kinetic energy of gas molecules.
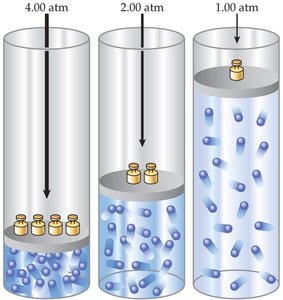
Boyle’s Law: Pressure–Volume Relationship
Boyle’s law states that at constant temperature, the volume of a gas is inversely proportional to its pressure.
Mathematical form:
Example: Compressing a gas in a piston increases its pressure and decreases its volume.
Charles’s Law: Temperature–Volume Relationship
Charles’s law states that at constant pressure, the volume of a gas is directly proportional to its absolute temperature (in Kelvin).
Mathematical form:
Example: A balloon expands when heated, as the gas inside increases in volume with temperature.


Avogadro’s Law: Volume–Amount Relationship
Avogadro’s law states that at constant temperature and pressure, the volume of a gas is directly proportional to the number of moles of gas present.
Mathematical form:
Standard Temperature and Pressure (STP): 0 °C (273.15 K) and 1 atm; at STP, 1 mole of any gas occupies 22.4 L.

Combined Gas Law
The combined gas law relates pressure, volume, and temperature for a fixed amount of gas.
Mathematical form:
Application: Used when a gas sample undergoes changes in pressure, volume, and temperature simultaneously.
Ideal Gas Law
The ideal gas law combines all the simple gas laws into one equation, relating pressure, volume, temperature, and amount of gas.
Mathematical form:
R (gas constant):
Application: Used to calculate any one property of a gas if the others are known.
Summary Table: Gas Laws
Law | Relationship | Equation |
|---|---|---|
Boyle's Law | Pressure & Volume (T constant) | |
Charles's Law | Volume & Temperature (P constant) | |
Avogadro's Law | Volume & Moles (P, T constant) | |
Combined Gas Law | P, V, T (n constant) | |
Ideal Gas Law | P, V, n, T |
Additional info: These notes expand on the original lecture outline by providing definitions, equations, and examples for each law and concept, as well as a summary table for quick reference.
