 Back
BackIntro to Chemistry Final Exam Study Guidance
Study Guide - Smart Notes

Q25. Write the complete electron configuration for phosphorus.
Background
Topic: Electron Configuration
This question tests your understanding of how electrons are arranged in an atom's orbitals, following the Aufbau principle, Pauli exclusion principle, and Hund's rule. Electron configurations are important for predicting chemical properties and reactivity.
Key Terms and Formulas
Electron configuration: The distribution of electrons among the atomic orbitals of an atom.
Aufbau principle: Electrons fill orbitals starting with the lowest energy first.
Pauli exclusion principle: Each orbital can hold a maximum of two electrons with opposite spins.
Hund's rule: Electrons fill degenerate orbitals singly before pairing up.
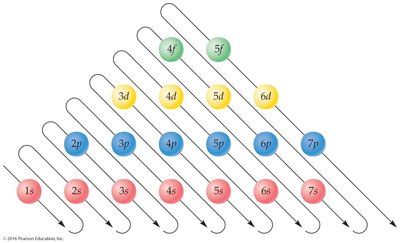
Step-by-Step Guidance
Find the atomic number of phosphorus (P). Phosphorus has atomic number 15, so it has 15 electrons.
Use the Aufbau diagram to determine the order in which orbitals are filled: .
Fill the orbitals in order, keeping track of the number of electrons in each:
can hold 2 electrons
can hold 2 electrons
can hold 6 electrons
can hold 2 electrons
can hold up to 6 electrons, but you only need to add enough to reach 15 total electrons
Write out the configuration as you fill each orbital, adding up the electrons as you go. Stop before the final step of completing the configuration.
Try solving on your own before revealing the answer!
Final Answer:
Phosphorus has 15 electrons, so the configuration fills up to .
This matches the order shown in the Aufbau diagram and the periodic table.
