 Back
BackExam 1
Study Guide - Smart Notes

Chapter 1: The Chemical World
Chemistry and Its Scope
Chemistry is the science that seeks to understand how matter behaves by studying the behavior of atoms and molecules. The arrangement and shape of molecules can dramatically affect the properties of substances.
Chemistry: The study of matter, its properties, and the changes it undergoes.
Molecules: Groups of atoms bonded together, whose structure determines the properties of substances.
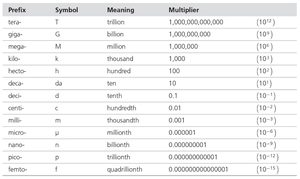
The Scientific Method
The scientific method is a systematic approach to learning about the natural world through observation and experimentation. It is characterized by the following steps:
Observation: Gathering data about nature.
Hypothesis: A tentative explanation for observations.
Experiment: Testing hypotheses through controlled procedures.
Law: A statement summarizing consistent experimental results (e.g., Law of Conservation of Mass).
Theory: A model that explains and unifies laws and observations.
Falsifiability: The potential for a hypothesis or theory to be proven wrong by experiment.
Example: The Law of Conservation of Mass states that in a chemical reaction, matter is neither created nor destroyed.
Chapter 2: Measurement and Problem Solving
Scientific Notation
Scientific notation expresses numbers as a product of a number between 1 and 10 and a power of ten. This is useful for very large or small numbers.
Move the decimal to create a number between 1 and 10.
Multiply by 10 raised to the number of places moved (positive for left, negative for right).
Significant Figures
Significant figures reflect the precision of a measured value. Rules for counting significant figures:
All nonzero digits are significant.
Zeros between nonzero digits are significant.
Leading zeros are not significant.
Trailing zeros are significant only if there is a decimal point.

Unit Conversions
Unit conversions use conversion factors to change from one unit to another. The dimensional analysis method ensures units cancel appropriately.

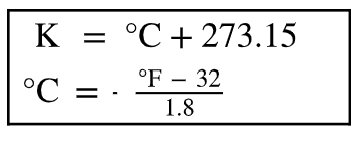

Temperature Conversions
Temperature can be measured in Celsius (°C), Kelvin (K), or Fahrenheit (°F). Conversion formulas are:

Density
Density is the ratio of mass to volume and is a key property for identifying substances.
Formula:
Chapter 3: Matter and Energy
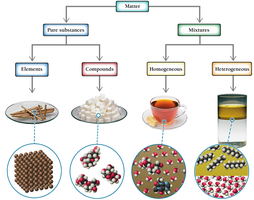
Classification of Matter
Matter is anything that occupies space and has mass. It can be classified as pure substances (elements and compounds) or mixtures (homogeneous and heterogeneous).
Element: A substance that cannot be broken down into simpler substances.
Compound: A substance composed of two or more elements in fixed proportions.
Homogeneous mixture: Uniform composition throughout (e.g., tea).
Heterogeneous mixture: Non-uniform composition (e.g., oil and water).

States of Matter
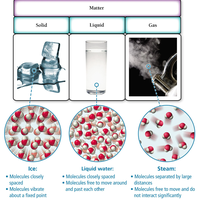
Matter exists in three primary states: solid, liquid, and gas. The arrangement and movement of molecules differ in each state.
Solid: Definite shape and volume; molecules vibrate in fixed positions.
Liquid: Definite volume, indefinite shape; molecules move past each other.
Gas: Indefinite shape and volume; molecules move freely and are far apart.
Physical and Chemical Properties
Physical properties can be observed without changing the substance's composition (e.g., color, melting point). Chemical properties are observed only when a substance undergoes a chemical change (e.g., flammability).
Physical and Chemical Changes
A physical change alters the form of a substance without changing its identity, while a chemical change produces a new substance.
Physical change: Melting ice, dissolving sugar in water.
Chemical change: Burning wood, rusting iron.
Energy and Its Forms
Energy is the capacity to do work. It exists in various forms, including kinetic, potential, thermal, electrical, and chemical energy.
Kinetic energy: Energy of motion.
Potential energy: Energy due to position or composition.
Thermal energy: Energy from the random motion of atoms and molecules.
Chemical energy: Stored in the bonds of chemical compounds.
Law of Conservation of Energy: Energy cannot be created or destroyed, only transformed.
Units of Energy
The SI unit of energy is the joule (J). Other common units include the calorie (cal) and kilowatt-hour (kWh).
1 calorie (cal) = 4.184 joules (J)
1 Calorie (Cal) = 1000 calories (cal)
1 kilowatt-hour (kWh) = joules (J)
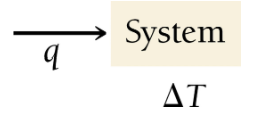
Heat, Temperature Change, and Specific Heat
Heat is the transfer of energy due to temperature difference. The amount of heat required to change the temperature of a substance depends on its mass, specific heat capacity, and the temperature change.
Formula:
q: heat (J), m: mass (g), C: specific heat capacity (J/g·°C), ΔT: temperature change (°C)


Example: Calculating the heat required to raise the temperature of water.
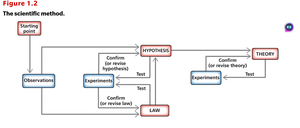
Summary Table: SI Prefixes
The following table summarizes common SI prefixes used in chemistry for expressing large and small quantities:
Prefix | Symbol | Meaning | Multiplier |
|---|---|---|---|
tera- | T | trillion | 1,000,000,000,000 (1012) |
giga- | G | billion | 1,000,000,000 (109) |
mega- | M | million | 1,000,000 (106) |
kilo- | k | thousand | 1,000 (103) |
hecto- | h | hundred | 100 (102) |
deca- | da | ten | 10 (101) |
deci- | d | tenth | 0.1 (10-1) |
centi- | c | hundredth | 0.01 (10-2) |
milli- | m | thousandth | 0.001 (10-3) |
micro- | μ | millionth | 0.000001 (10-6) |
nano- | n | billionth | 0.000000001 (10-9) |
pico- | p | trillionth | 0.000000000001 (10-12) |
femto- | f | quadrillionth | 0.000000000000001 (10-15) |

Summary Table: Common Unit Conversions
Quantity | Conversion |
|---|---|
Length | 1 kilometer (km) = 0.6214 mile (mi) 1 meter (m) = 39.37 inches (in.) 1 foot (ft) = 1.094 yards (yd) 1 inch (in.) = 2.54 centimeters (cm) (exact) |
Mass | 1 kilogram (kg) = 2.205 pounds (lb) 1 pound (lb) = 453.59 grams (g) 1 ounce (oz) = 28.35 grams (g) |
Volume | 1 liter (L) = 1000 milliliters (mL) = 1000 cubic centimeters (cm3) 1 liter (L) = 1.057 quarts (qt) 1 U.S. gallon (gal) = 3.785 liters (L) |

