 Back
BackLiquids and Solids: Intermolecular Forces, Properties, and Structures
Study Guide - Smart Notes

Liquids and Solids
Introduction to Liquids and Solids
The properties of liquids and solids are fundamentally determined by the types and strengths of intermolecular forces (IMFs) present. Unlike gases, where properties are largely independent of chemical identity, the behavior of liquids and solids is closely tied to the nature of the constituent particles and their interactions.
Intermolecular Forces (IMFs)
Types of Intermolecular Forces
Intermolecular forces are the noncovalent attractions between molecules or atoms in condensed phases. They are much weaker than the covalent (intramolecular) bonds holding atoms together within a molecule, but they are crucial in determining physical properties such as boiling and melting points, viscosity, and solubility.
Dispersion Forces (London Dispersion Forces): Present in all substances, these arise from temporary fluctuations in electron distribution, creating instantaneous dipoles that induce dipoles in neighboring particles.
Dipole-Dipole Attractions: Occur in polar molecules with permanent dipoles, aligning so that the positive end of one molecule is near the negative end of another.
Hydrogen Bonding: A special, strong type of dipole-dipole interaction found when hydrogen is covalently bonded to highly electronegative atoms (N, O, or F).
These forces are collectively known as van der Waals forces.
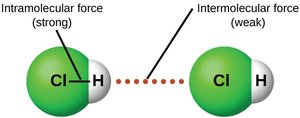
Comparison of Intermolecular and Intramolecular Forces
Intramolecular forces (covalent bonds) are much stronger than intermolecular forces. The differences in the strengths of IMFs account for the different physical states (solid, liquid, gas) and their transitions.
Dispersion Forces and Molar Mass
Dispersion forces increase with the size and mass of the molecule due to greater polarizability. Larger atoms have more easily distorted electron clouds, leading to stronger dispersion forces and higher boiling/melting points.
Dipole-Dipole Attractions and Boiling Points
Polar molecules experience both dispersion and dipole-dipole forces. The presence of dipole-dipole attractions leads to higher boiling points compared to nonpolar molecules of similar molar mass.
Hydrogen Bonding
Hydrogen bonding is the strongest van der Waals force and is responsible for many unique properties of substances like water and biological molecules such as DNA. It occurs only when hydrogen is bonded to N, O, or F, resulting in highly concentrated partial charges and strong attractions.
Boiling Point Trends in Hydrides
Hydrogen bonding causes anomalously high boiling points for hydrides of period 2 elements (HF, H2O, NH3) compared to heavier congeners, as shown in the following graphs:
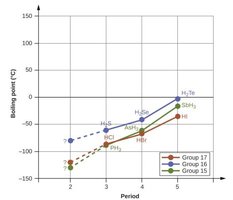
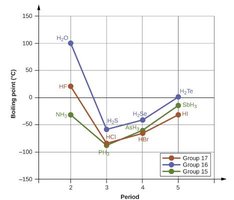
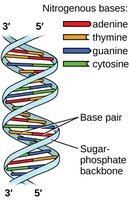
Hydrogen Bonding in Biological Molecules
Hydrogen bonds play a critical role in stabilizing the double helix structure of DNA by holding complementary base pairs together.
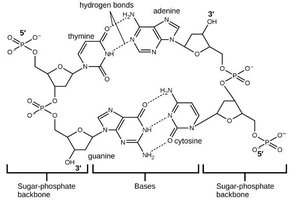
Properties of Liquids
Cohesive and Adhesive Forces
Cohesive forces are IMFs between like molecules, while adhesive forces are IMFs between unlike molecules. These forces explain phenomena such as surface tension, viscosity, and capillary action.
Viscosity
Viscosity is a measure of a liquid's resistance to flow. Substances with strong IMFs (e.g., honey, motor oil) have high viscosities.
Surface Tension
Surface tension is the energy required to increase the surface area of a liquid. It results from cohesive forces at the liquid's surface, allowing phenomena such as water droplets forming spheres and insects walking on water.
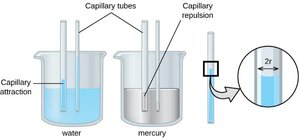
Capillary Action
Capillary action is the movement of liquid within narrow spaces due to adhesive and cohesive forces. The shape of the meniscus in a tube depends on the relative strengths of these forces.
Capillary action is used in medical applications, such as drawing blood into capillary tubes.
Phase Transitions
Phase Changes and Equilibrium
Phase transitions include vaporization, condensation, melting, freezing, sublimation, and deposition. At equilibrium, the rate of phase change in one direction equals the rate in the opposite direction (e.g., liquid ⇌ vapor).
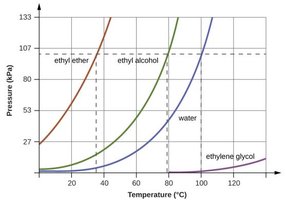
Vapor Pressure
Vapor pressure is the pressure exerted by a vapor in equilibrium with its liquid. It depends on temperature and the strength of IMFs; substances with weaker IMFs have higher vapor pressures at a given temperature.
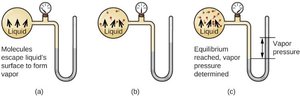
Temperature Dependence of Vapor Pressure
As temperature increases, more molecules have sufficient kinetic energy to escape the liquid phase, increasing vapor pressure.
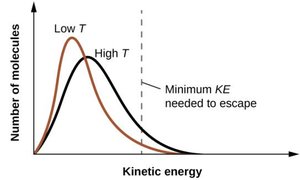
Boiling Point and External Pressure
The boiling point is the temperature at which vapor pressure equals external pressure. The normal boiling point is measured at 1 atm. Boiling points decrease at higher altitudes (lower pressure).
Heating Curves and Phase Changes
During a phase change, temperature remains constant as energy is used to break or form IMFs rather than changing kinetic energy.

Phase Diagrams
Reading Phase Diagrams
Phase diagrams graphically represent the stable phases of a substance at various temperatures and pressures. They show regions for solid, liquid, gas, and sometimes supercritical fluid phases, as well as lines of equilibrium (phase boundaries).
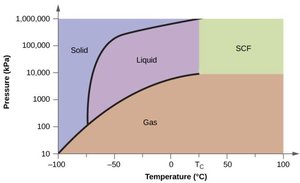
Supercritical Fluids
Above the critical temperature and pressure, a substance forms a supercritical fluid, which has properties of both liquids and gases. Supercritical CO2 is used in processes such as decaffeination of coffee.
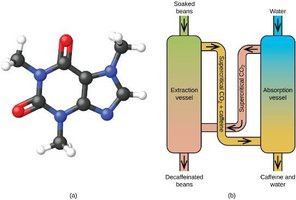
The Solid State of Matter
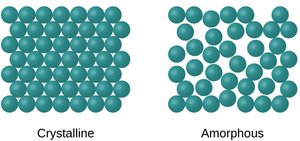
Crystalline vs. Amorphous Solids
Solids can be classified as crystalline (ordered, repeating patterns) or amorphous (disordered, no long-range order).
Types of Crystalline Solids
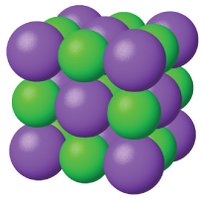
Ionic solids: Composed of cations and anions held together by electrostatic forces (e.g., NaCl).
Metallic solids: Metal atoms in a sea of delocalized electrons (e.g., copper).
Covalent network solids: Atoms connected by a continuous network of covalent bonds (e.g., diamond, SiO2).
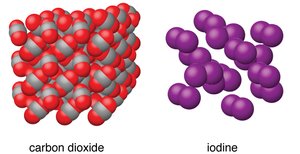
Molecular solids: Molecules held together by IMFs (e.g., CO2, I2).
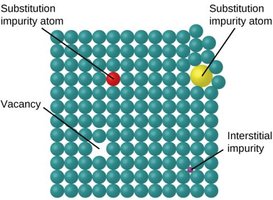
Crystal Defects
Crystalline solids may contain defects such as vacancies (missing atoms), interstitial atoms (extra atoms in spaces), and substitutional impurities (foreign atoms replacing host atoms).
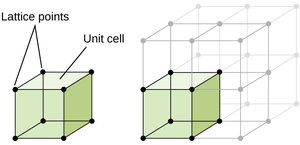
Lattice Structures in Crystalline Solids
Unit Cells and Lattice Points
The unit cell is the smallest repeating unit in a crystal lattice. Lattice points represent the positions of atoms, ions, or molecules in the structure.
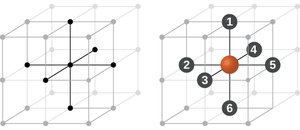
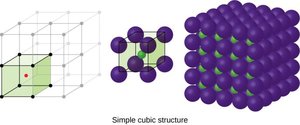
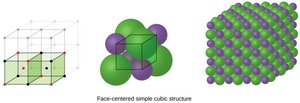
Cubic Lattice Structures
There are several types of cubic unit cells:
Simple cubic (SC): Atoms at the corners only; coordination number 6.
Body-centered cubic (BCC): Atoms at corners and one in the center; coordination number 8.
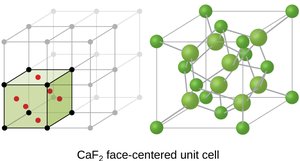
Face-centered cubic (FCC): Atoms at corners and centers of faces; coordination number 12.
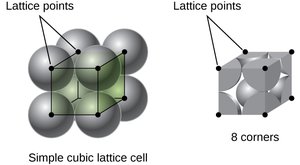
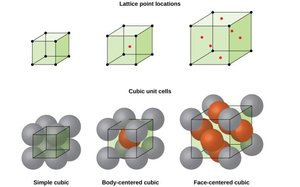
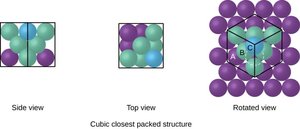
Lattice Systems and Unit Cells
There are seven crystal systems and 14 Bravais lattices, each defined by the lengths and angles of the unit cell axes.
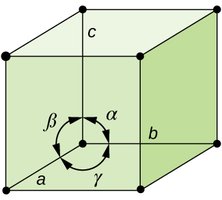

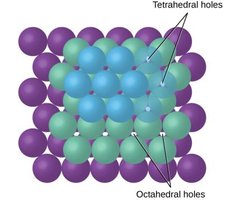
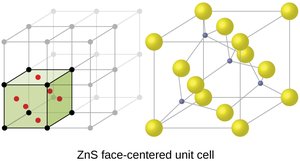
Ionic Structures and Holes
Cations in ionic solids may occupy octahedral or tetrahedral holes between anions, depending on their relative sizes.
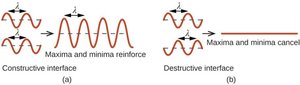
X-ray Diffraction and Crystal Structure
X-ray diffraction is a key technique for determining the arrangement of atoms in a crystal. When X-rays interact with a crystal, they produce a diffraction pattern that can be analyzed to reveal the structure.
Additional info: This summary covers the main topics and subtopics from the provided materials, expanding on definitions, examples, and applications relevant to an introductory college chemistry course.
