 Back
BackLiquids, Solids, and Intermolecular Forces: Structured Study Notes
Study Guide - Smart Notes

Liquids, Solids, and Intermolecular Forces
Interactions Between Molecules
Intermolecular forces are the attractive forces that exist among molecules and atoms, playing a crucial role in determining the physical state of matter. These forces are responsible for the formation of liquids and solids, as well as many physiological processes in living organisms.

States of Matter: Solid, Liquid, and Gas
The physical state of a substance—solid, liquid, or gas—is determined by the balance between intermolecular forces and thermal energy. Molecules and atoms are in constant motion, and their arrangement and movement differ in each state.
Solid: Molecules are closely packed and fixed in position, vibrating about a fixed point.
Liquid: Molecules are in close contact but can move around each other, allowing flow.
Gas: Molecules are far apart and move freely, filling the container.

Properties of Gases, Liquids, and Solids
Each state of matter exhibits distinct properties due to differences in intermolecular forces:
Gases: Low density, indefinite shape and volume, easily compressed, weak intermolecular forces.
Liquids: High density (compared to gases), indefinite shape, definite volume, not easily compressed, moderate intermolecular forces.
Solids: High density, definite shape and volume, not easily compressed, strong intermolecular forces, may be crystalline or amorphous.
Shape and Movement in Liquids and Solids
Liquids flow and assume the shape of their container due to the freedom of movement of their molecules. In contrast, solids have molecules fixed in place, though they vibrate about their positions.

Manifestations of Intermolecular Forces: Surface Tension and Viscosity
Intermolecular forces are responsible for phenomena such as surface tension and viscosity in liquids.
Surface Tension: The tendency of liquids to minimize their surface area, creating a "skin" that resists penetration.
Viscosity: The resistance of a liquid to flow; liquids with strong intermolecular forces or long molecules are more viscous.

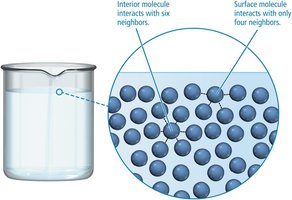

Evaporation and Condensation
Evaporation is the process by which molecules at the surface of a liquid gain enough energy to escape into the gas phase. The rate of evaporation increases with surface area, temperature, and decreases with stronger intermolecular forces. Condensation is the reverse process, where gas molecules return to the liquid state. When the rates of evaporation and condensation are equal, dynamic equilibrium is reached, and the vapor pressure becomes constant.
Volatile liquids: Evaporate easily.
Nonvolatile liquids: Do not evaporate easily.
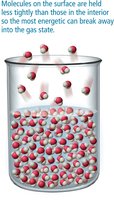
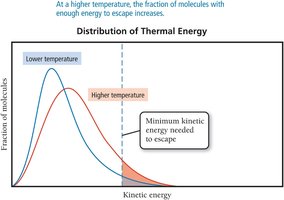
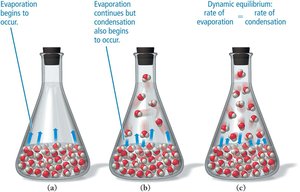
Vapor Pressure and Boiling
Vapor pressure is the partial pressure of a vapor in equilibrium with its liquid. It increases with temperature and decreases with stronger intermolecular forces. Boiling occurs when the vapor pressure equals the external pressure, allowing molecules throughout the liquid to vaporize.
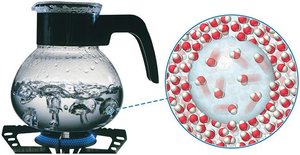
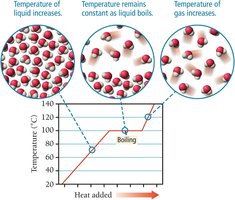
Energetics of Phase Changes
Phase changes involve energy transfer:
Evaporation: Endothermic; absorbs heat.
Condensation: Exothermic; releases heat.
Melting: Endothermic; absorbs heat.
Freezing: Exothermic; releases heat.
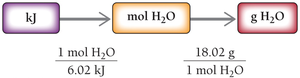
Heat of Vaporization
The heat of vaporization () is the amount of heat required to vaporize 1 mole of liquid. For water at 100 ºC, .
Formula:

Heat of Fusion
The heat of fusion () is the amount of heat required to melt 1 mole of solid. For water, .
Formula:

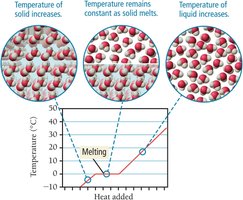
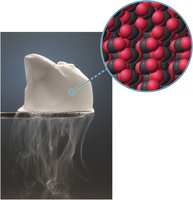
Sublimation
Sublimation is the direct transition from solid to gas without passing through the liquid phase. Dry ice (solid CO2) sublimes at atmospheric pressure.
Types of Intermolecular Forces
Intermolecular forces determine the physical properties of substances. The four main types are:
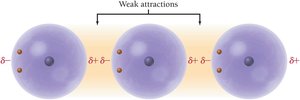
Dispersion Forces (London Forces): Present in all molecules and atoms due to temporary fluctuations in electron distribution.
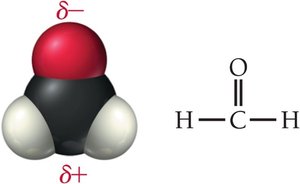
Dipole–Dipole Forces: Occur in polar molecules with permanent dipoles.
Hydrogen Bonding: Occurs in molecules with hydrogen bonded directly to F, O, or N; strongest among the three.
Ion–Dipole Forces: Occur in mixtures of ionic and polar compounds.

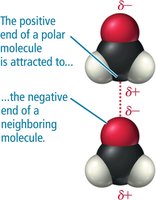
Polarity and Miscibility
Miscibility refers to the ability of liquids to mix without separating. Polar liquids mix with other polar liquids, but not with nonpolar liquids. Water (polar) does not mix with pentane or oil (nonpolar).

Determining Dipole–Dipole Forces
To determine if a molecule has dipole–dipole forces:
Check for polar bonds.
Determine if the polar bonds add up to a net dipole moment.
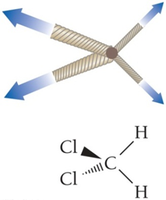
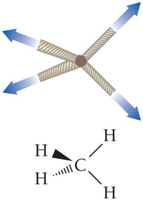
Hydrogen Bonding
Hydrogen bonding occurs in molecules where hydrogen is bonded directly to fluorine, oxygen, or nitrogen. This results in a strong attraction between molecules, significantly raising melting and boiling points.
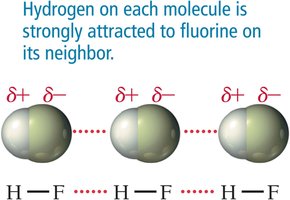
Types of Crystalline Solids
Crystalline solids are classified based on their composite units:
Molecular Solids: Composite units are molecules; held together by intermolecular forces.
Ionic Solids: Composite units are formula units (cations and anions); held together by ionic bonds.
Atomic Solids: Composite units are atoms; held together by covalent, metallic, or dispersion forces.
Water: A Remarkable Molecule
Water is unique due to its strong hydrogen bonding, high polarity, and ability to dissolve many substances. It has a high boiling point for its molar mass and expands upon freezing, making ice less dense than liquid water.
Summary Table: Types of Intermolecular Forces
Type | Occurs In | Relative Strength |
|---|---|---|
Dispersion | All molecules/atoms | Weak |
Dipole–Dipole | Polar molecules | Moderate |
Hydrogen Bond | H bonded to F, O, N | Strong |
Ion–Dipole | Ionic + polar mixtures | Very strong |
Key Equations
Heat of Vaporization:
Heat of Fusion:
Learning Objectives
Describe properties of solids and liquids and relate them to their constituent atoms and molecules.
Explain surface tension and viscosity as manifestations of intermolecular forces.
Describe and explain evaporation, condensation, melting, freezing, and sublimation.
Use heats of vaporization and fusion in calculations.
Compare and contrast types of intermolecular forces.
Identify types of crystalline solids.
Describe the unique properties of water.
