 Back
BackMatter and Energy: Classification, States, and Properties
Study Guide - Smart Notes

Matter and Energy
Classification of Matter
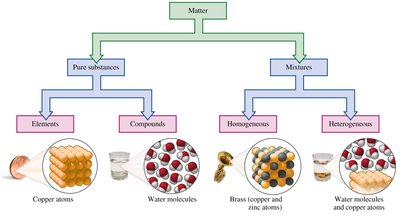
Matter is defined as anything that has mass and occupies space. It can be classified based on its composition into pure substances and mixtures.
Pure Substances: These have a fixed or definite composition and can be further classified as elements or compounds.
Mixtures: These consist of two or more substances physically mixed, not chemically combined, and can be separated by physical methods.

Elements and Compounds
Elements are composed of only one type of atom and cannot be broken down further by chemical means. Compounds consist of atoms of two or more elements chemically combined in a fixed proportion and can be broken down into simpler substances by chemical processes.
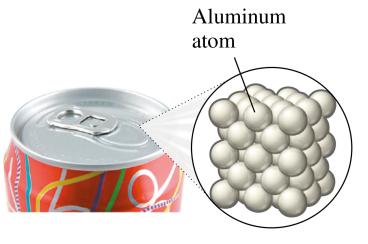
Example of Element: Aluminum in a soda can is composed of aluminum atoms.
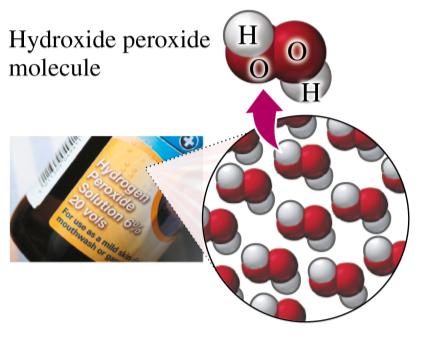
Example of Compound: Hydrogen peroxide solution contains molecules made of hydrogen and oxygen atoms.
Mixtures: Homogeneous and Heterogeneous
Mixtures can be classified as homogeneous (uniform composition, also called solutions) or heterogeneous (non-uniform composition, distinct phases present).
Homogeneous Mixture Example: Brass (copper and zinc atoms), air, seawater.
Heterogeneous Mixture Example: Oil and water, salad, soda.

States and Properties of Matter
Matter exists in three primary states: solid, liquid, and gas. Each state has distinct properties regarding shape, volume, particle arrangement, and compressibility.
Solid: Definite shape and volume; particles are closely packed and rigid. Compressibility is very slight.
Liquid: Definite volume but no definite shape; particles are close together but move freely. Slightly compressible.
Gas: Indefinite shape and volume; particles are far apart and move independently. Highly compressible.

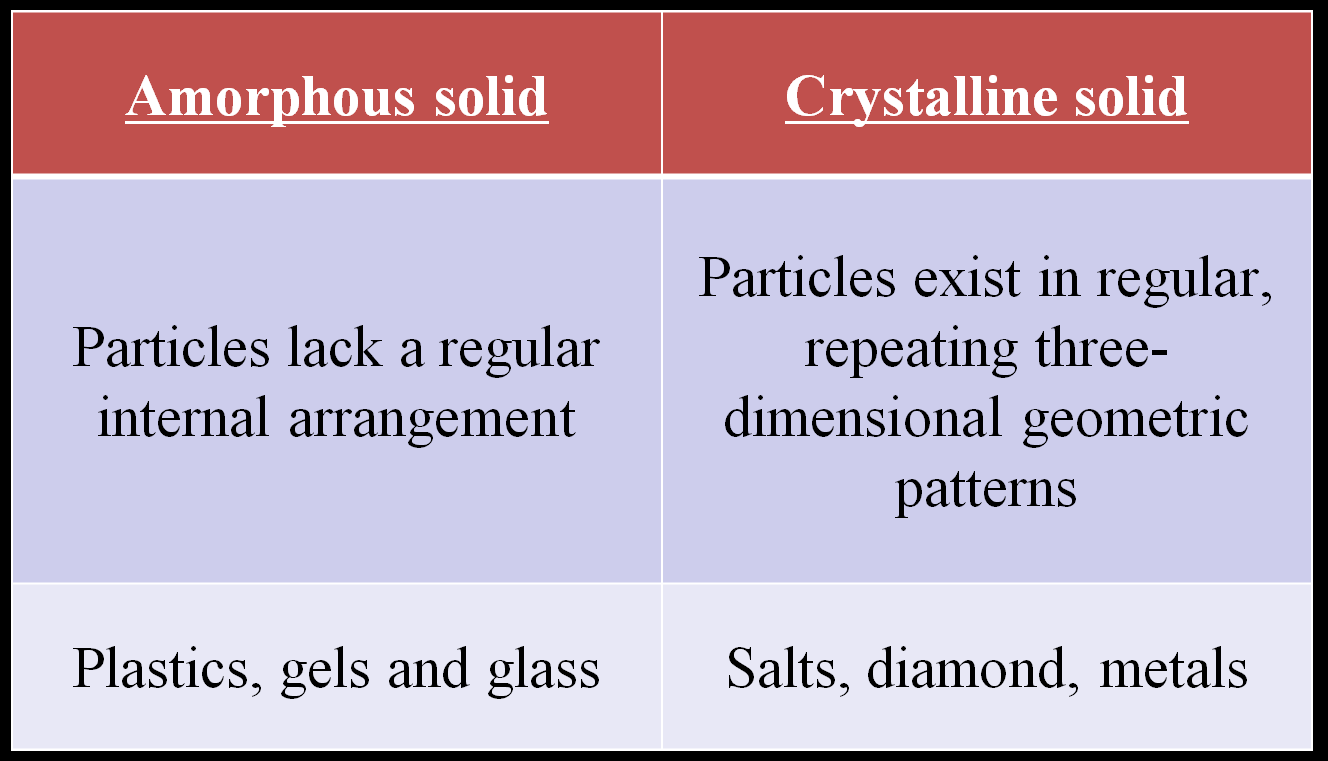
Types of Solids
Solids can be classified as amorphous or crystalline based on the arrangement of their particles.
Amorphous solid | Crystalline solid |
|---|---|
Particles lack a regular internal arrangement | Particles exist in regular, repeating three-dimensional geometric patterns |
Plastics, gels, and glass | Salts, diamond, metals |
Properties of Matter
Physical Properties and Changes
Physical properties are inherent characteristics that can be observed or measured without changing the substance's composition. Physical changes alter physical properties or the state of matter but do not change the substance's identity.
Examples of Physical Properties: Taste, color, odor, size, shape, luster, density, melting point, boiling point.
Examples of Physical Changes: Tearing paper, melting ice, boiling water, dissolving sugar in water.

Chemical Properties and Changes
Chemical properties describe a substance's ability to form new substances by reaction or decomposition. Chemical changes result in the formation of new substances with different properties and composition.
Examples of Chemical Changes: Burning paper, rusting iron, heating sugar to form caramel.

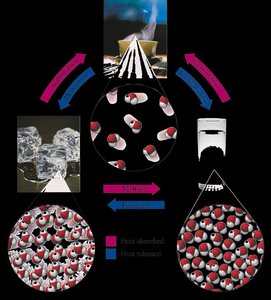
Energy and Changes of State
Energy is the capacity of matter to do work. It exists in various forms, including mechanical, chemical, electrical, heat, nuclear, and radiant energy. Matter undergoes changes of state (solid, liquid, gas) through the absorption or release of energy.
Kinetic Energy: Energy due to motion.
Potential Energy: Stored energy due to position or chemical bonds.
Heat: A form of energy associated with the motion of particles; also known as thermal energy.
Temperature: A measure of the intensity of heat.
Summary Table: Physical vs. Chemical Changes
Physical Changes | Chemical Changes |
|---|---|
Water boils to form water vapor | Water and cesium combine explosively |
Paper is cut into tiny pieces of confetti | Paper burns with a bright flame and produces heat, ashes, carbon dioxide, and water vapor |
Sugar dissolves in water to form a sugar solution | Heating sugar forms a smooth, caramel-colored substance |
Iron changes from a solid to a liquid at 1538 °C | Iron combines with oxygen to form orange-red iron oxide (rust) |


Additional info:
These notes cover the fundamental concepts of matter and energy, including classification, states, properties, and changes. The included images and tables reinforce the classification of matter, the differences between physical and chemical changes, and the transitions between states of matter.
