 Back
BackNomenclature of Ionic and Covalent Compounds: Study Guide
Study Guide - Smart Notes

Nomenclature of Ionic and Covalent Compounds
Naming Ionic Compounds
Ionic compounds are formed from the combination of metals and nonmetals. The nomenclature follows a systematic approach to ensure clarity and consistency.
Metal Name: The name of the metal (cation) is written first and remains unchanged from its elemental name.
Nonmetal Name: The name of the nonmetal (anion) is written second, using the first syllable of its name followed by the suffix -ide.
Spacing: A space separates the names of the metal and nonmetal ions.
Example: K2O is named potassium oxide.
Metals with Variable Charge
Many transition metals can form more than one type of positive ion (cation). Their charge is specified using Roman numerals in parentheses immediately after the metal name.
Examples: Cu2+ is copper(II), Cu+ is copper(I), Fe2+ is iron(II), Fe3+ is iron(III).
Exceptions: Zn2+, Cd2+, and Ag+ always have a fixed charge and do not require Roman numerals.
Determining Variable Charge
The charge of a variable-charge metal ion is determined by balancing the charges of the cation and anion in the compound.
Example: In MnF2, manganese must be Mn2+ to balance two F- ions.
Formula:
Ion Charges and the Periodic Table
The periodic table helps predict the charges of ions formed by elements. Metals typically form positive ions, while nonmetals form negative ions.
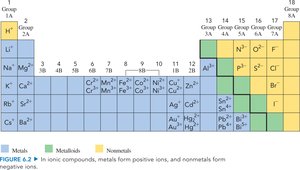
Naming Ionic Compounds with Variable Charge Metals
When naming compounds with variable-charge metals, the charge is determined from the anion and indicated with a Roman numeral.
Example: FeCl2 is named iron(II) chloride.
Steps: Determine cation charge, name cation with Roman numeral, name anion with -ide, write cation first.
Writing Formulas from Compound Names
To write the formula for an ionic compound, balance the charges of the cation and anion, then use subscripts to indicate the number of each ion.
Example: Iron(III) chloride: Fe3+ and Cl- combine as FeCl3.
Formula:
Polyatomic Ions
Polyatomic ions are groups of atoms with an overall charge, often containing a nonmetal and oxygen. Most are negatively charged, except ammonium (NH4+) and hydronium (H3O+).
Common Polyatomic Ions: sulfate (SO42-), nitrate (NO3-), phosphate (PO43-), etc.
Exceptions: cyanide (CN-), hydroxide (OH-).
Naming Polyatomic Ions
The names of polyatomic ions follow specific rules based on their composition:
-ate: Most common form (e.g., sulfate, nitrate).
-ite: One fewer oxygen atom (e.g., sulfite, nitrite).
Adding H+: Forms hydrogen carbonate (bicarbonate), hydrogen sulfate (bisulfate), etc.
Anions Containing Hydrogen
Some polyatomic anions contain hydrogen, which affects their name and charge.
Examples: HCO3- (bicarbonate), H2PO4- (dihydrogen phosphate), HSO4- (hydrogen sulfate).
Naming Acids
Acids are named based on the anion they contain. If the anion does not contain oxygen, use the prefix hydro- and the suffix -ic acid. If the anion contains oxygen, use -ic or -ous depending on the ending of the anion name.
-ate anion: Use -ic acid (e.g., sulfate → sulfuric acid).
-ite anion: Use -ous acid (e.g., sulfite → sulfurous acid).
Writing Formulas for Compounds with Polyatomic Ions
Formulas for compounds containing polyatomic ions are written by balancing charges, using parentheses around the polyatomic ion if more than one is needed.
Example: Magnesium nitrate: Mg2+ and NO3- combine as Mg(NO3)2.
Formula:
Writing Formulas with Polyatomic Ions: Aluminum Bicarbonate
To write the formula for aluminum bicarbonate, balance the charges and use parentheses for the polyatomic ion.
Example: Al3+ and HCO3- combine as Al(HCO3)3.
Formula:
Naming Compounds with Polyatomic Ions
When naming compounds containing polyatomic ions, write the positive ion first (usually a metal), followed by the name of the polyatomic ion. No prefixes are used.

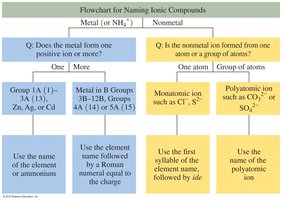
Flowchart for Naming Ionic Compounds
A flowchart can help determine the correct naming procedure for ionic compounds, based on whether the metal forms one or more positive ions and whether the nonmetal is a monoatomic or polyatomic ion.
Binary Covalent Compounds
Binary covalent compounds are formed between two nonmetals. The naming uses prefixes to indicate the number of each atom present.
First Element: Name as is.
Second Element: Name as an anion (with -ide suffix).
Prefixes: Indicate the number of atoms (mono-, di-, tri-, tetra-, etc.). Do not use 'mono-' for the first element.
Example: SiF4 is named silicon tetrafluoride.
Greek Prefixes for Covalent Compounds
mono-: 1
di-: 2
tri-: 3
tetra-: 4
penta-: 5
hexa-: 6
hepta-: 7
octa-: 8
Nomenclature Practice Examples
SiF4: Silicon tetrafluoride
CuNO2: Copper(I) nitrite
Cl2O7: Dichlorine heptoxide
H2SO3: Sulfurous acid
NaHCO3: Sodium bicarbonate
AsCl3: Arsenic trichloride
CaS: Calcium sulfide
HClO4: Perchloric acid
AgOH: Silver hydroxide
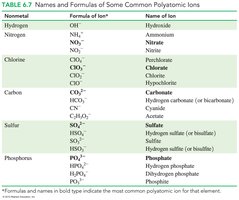
Table: Names and Formulas of Common Polyatomic Ions
This table summarizes the most common polyatomic ions, their formulas, and their names.
Nonmetal | Formula of Ion | Name of Ion |
|---|---|---|
Hydrogen | OH- | Hydroxide |
Hydrogen | NH4+ | Ammonium |
Nitrogen | NO3- | Nitrate |
Nitrogen | NO2- | Nitrite |
Chlorine | ClO4- | Perchlorate |
Chlorine | ClO3- | Chlorate |
Chlorine | ClO2- | Chlorite |
Chlorine | ClO- | Hypochlorite |
Carbon | CO32- | Carbonate |
Carbon | HCO3- | Hydrogen carbonate (bicarbonate) |
Carbon | CN- | Cyanide |
Carbon | C2H3O2- | Acetate |
Sulfur | SO42- | Sulfate |
Sulfur | HSO4- | Hydrogen sulfate (bisulfate) |
Sulfur | SO32- | Sulfite |
Sulfur | HSO3- | Hydrogen sulfite (bisulfite) |
Phosphorus | PO43- | Phosphate |
Phosphorus | HPO42- | Hydrogen phosphate |
Phosphorus | H2PO4- | Dihydrogen phosphate |
