Back
BackOrganic Chemistry: Structure, Properties, and Functional Groups
Study Guide - Smart Notes

Organic Chemistry
Definition and Scope
Organic chemistry is the branch of chemistry that studies hydrocarbons and their derivatives. Over 95% of known chemical compounds are carbon-based, making organic chemistry a central field in science.
Hydrocarbons: Compounds composed entirely of carbon and hydrogen.
Derivatives: Compounds formed by substituting other atoms or groups for hydrogen in hydrocarbons.
The Unique Carbon Atom
Bonding and Structure
Carbon atoms can bond to each other, forming long chains and rings, which is the basis for the diversity of organic compounds.
Catenation: The ability of carbon to form stable bonds with itself.
Versatility: Enables the formation of a wide variety of structures, including straight chains, branched chains, and rings.
Hydrocarbons
Classification
Hydrocarbons are classified based on the types of bonds between carbon atoms.
Alkanes: Only single bonds (saturated hydrocarbons).
Alkenes: At least one double bond (unsaturated hydrocarbons).
Alkynes: At least one triple bond (unsaturated hydrocarbons).
Alkanes
Structure and Nomenclature
Alkanes are saturated hydrocarbons with only single bonds. Their general formula is .
Naming: Prefix indicates the number of carbon atoms, suffix is '-ane'.
Examples: Methane (), Ethane (), Propane ().
Condensed Structural Formula: Shows the arrangement of atoms without drawing all bonds.
Stems for Organic Molecule Names
Stem | Number of Carbon Atoms |
|---|---|
Meth- | 1 |
Eth- | 2 |
Prop- | 3 |
But- | 4 |
Pent- | 5 |
Hex- | 6 |
Hept- | 7 |
Oct- | 8 |
Non- | 9 |
Dec- | 10 |
Homologous Series and Isomerism
Alkanes form a homologous series, differing by a group. Isomers have the same molecular formula but different structural arrangements.
Isomerism: Structural isomers differ in connectivity.
Physical Properties, Uses, and Occurrences of Selected Alkanes
Name | Molecular Formula | Melting Point (°C) | Boiling Point (°C) | Density at 20°C (g/mL) | Use/Occurrence |
|---|---|---|---|---|---|
Methane | CH4 | -183 | -162 | Gas | Natural gas (main component); fuel |
Ethane | C2H6 | -172 | -89 | Gas | Natural gas (minor component); plastics |
Propane | C3H8 | -188 | -42 | Gas | LPG (bottled gas); plastics |
Butane | C4H10 | -138 | 0 | Gas | LPG; lighter fuel |
Pentane | C5H12 | -130 | 36 | 0.626 | Gasoline component |
Hexane | C6H14 | -95 | 69 | 0.659 | Gasoline component; extraction solvent for food oils |
Heptane | C7H16 | -91 | 98 | 0.684 | Gasoline component |
Octane | C8H18 | -57 | 126 | 0.703 | Gasoline component |
Cyclic Hydrocarbons
Structure and Nomenclature
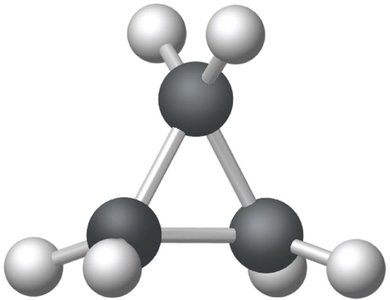
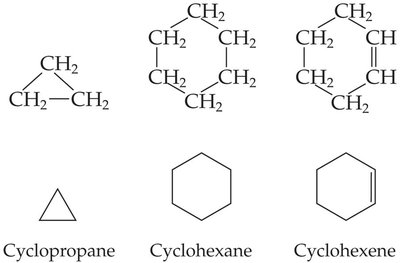
Cyclic hydrocarbons are ring compounds. The simplest is cyclopropane. The prefix 'cyclo-' is used in naming.
Unsaturated Hydrocarbons
Alkenes
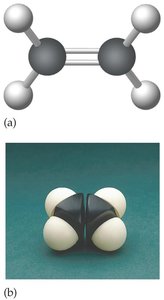
Alkenes contain at least one carbon-to-carbon double bond. Their general formula is , and their names end with '-ene'.
Example: Ethylene ()
Alkynes
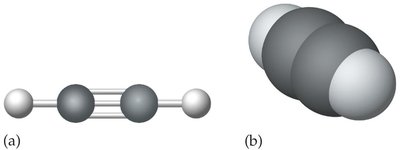
Alkynes contain at least one carbon-to-carbon triple bond. Their general formula is , and their names end with '-yne'.
Example: Ethyne (acetylene, )
Cis–Trans Isomerism
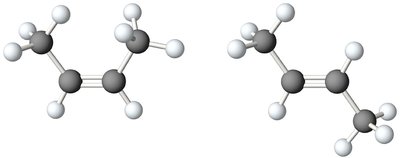
Certain alkenes and cyclic hydrocarbons can exist as cis–trans isomers, a form of geometric isomerism where isomers have the same connectivity but differ in spatial orientation.
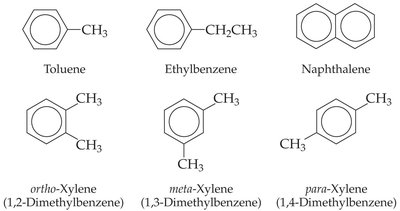
Aromatic Hydrocarbons
Benzene and Relatives
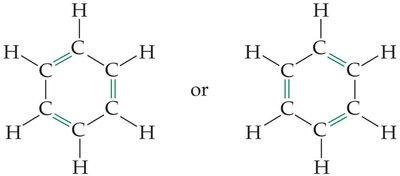
Benzene is a stable six-carbon ring. Aromatic hydrocarbons contain a benzene ring or have similar properties.
Chlorinated Hydrocarbons
Structure and Uses
Chlorinated hydrocarbons are formed by substituting chlorine for hydrogen in hydrocarbons. They are used as solvents, paint removers, and formerly as anesthetics.
Dichloromethane: Solvent, paint remover
Trichloromethane (chloroform): Solvent, hazardous anesthetic
Functional Groups
Definition and Importance
Functional groups are atoms or groups of atoms attached to hydrocarbon skeletons, giving compounds characteristic properties. Examples include double/triple bonds and halogen substituents.
Selected Organic Functional Groups
Name of Class | Functional Group | General Formula |
|---|---|---|
Alkane | None | R-H |
Alkene | C=C | R2C=CR2 |
Alkyne | C≡C | RC≡CR |
Alcohol | –OH | R–OH |
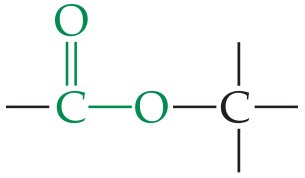
Ether | –O– | R–O–R' |
Aldehyde | –CHO | R–CHO |
Ketone | –CO– | R–CO–R' |
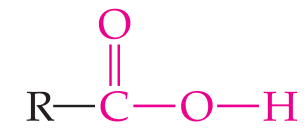
Carboxylic acid | –COOH | R–COOH |
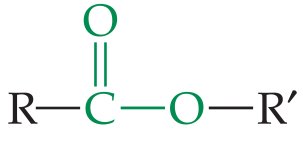
Ester | –COOR' | R–COOR' |
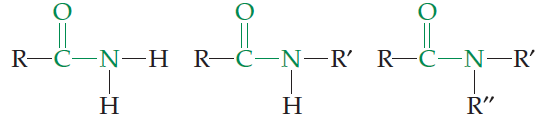
Amine | –NH2, –NHR, –NR2 | R–NH2, R–NHR, R–NR2 |
Amide | –CONH2, –CONHR, –CONR2 | R–CONH2, R–CONHR, R–CONR2 |
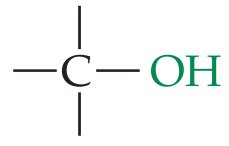
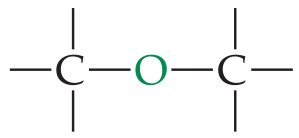
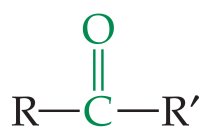
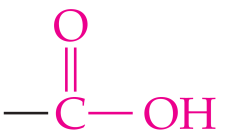
Alcohols
Structure and Examples
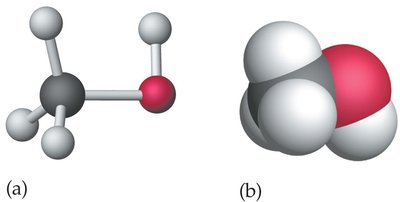
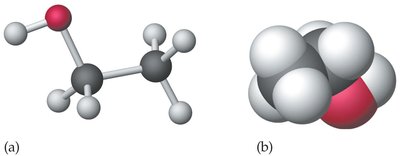
Alcohols contain the hydroxyl (–OH) functional group. Common examples include methanol, ethanol, and 1-propanol.
Methanol:
Ethanol:
1-Propanol:
Toxicity of Alcohols
All alcohols are toxic. Methanol is oxidized to formaldehyde, which is poisonous. Ethanol's toxicity causes inebriety, alcoholism, and fetal alcohol syndrome.
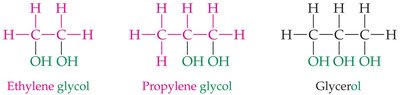
Multifunctional Alcohols
Some alcohols contain more than one hydroxyl group, such as ethylene glycol and glycerol.
Phenols
Structure and Properties
Phenols are aromatic compounds with a hydroxyl group attached to the ring. They act as acids and are effective antiseptics.
Ethers
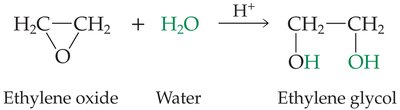
Structure and Examples
Ethers have two alkyl groups bonded to the same oxygen atom. The general formula is or .
Example: Diethyl ether ()
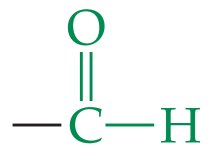
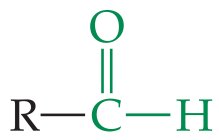
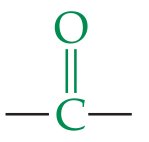
Aldehydes and Ketones
Structure and Functional Group
Both contain the carbonyl group (). Aldehydes have the group at the end of a chain, ketones have it within the chain.
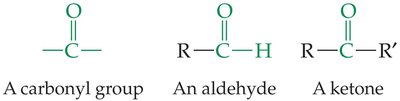
Carboxylic Acids
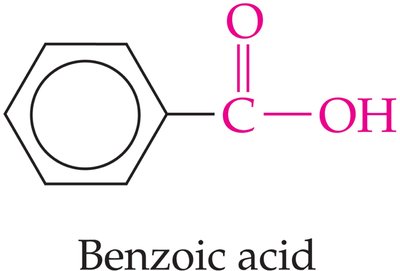
Structure and Properties
Carboxylic acids contain the carboxyl group (). When attached to a benzene ring, the compound is called benzoic acid.
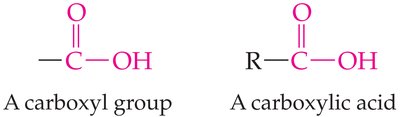
Esters
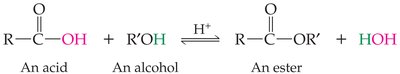
Formation and Properties
Esters are formed by replacing the hydrogen of a carboxylic acid with the alkyl group of an alcohol or phenol. They often have pleasant odors.
Ester Flavors and Fragrances
Ester | Formula | Flavor/Fragrance |
|---|---|---|
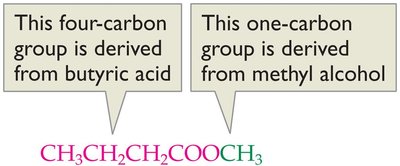
Methyl butyrate | CH3CH2CH2COOCH3 | Apple |
Ethyl butyrate | CH3CH2CH2COOCH2CH3 | Pineapple |
Propyl acetate | CH3COOCH2CH2CH3 | Pear |
Pentyl acetate | CH3COOCH2CH2CH2CH2CH3 | Banana |
Pentyl butyrate | CH3CH2CH2COOCH2CH2CH2CH2CH3 | Apricot |
Octyl acetate | CH3COOCH2CH2CH2CH2CH2CH2CH2CH3 | Orange |
Methyl benzoate | C6H5COOCH3 | Kiwifruit |
Ethyl formate | HCOOCH2CH3 | Rum |
Methyl salicylate | o-HOC6H4COOCH3 | Wintergreen |
Benzyl acetate | CH3COOCH2C6H5 | Jasmine |
Amines and Amides
Amines
Amines are derivatives of ammonia, formed by replacing one or more hydrogens with alkyl groups. They are generally basic and have distinctive odors.
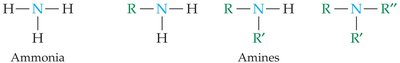
Amides
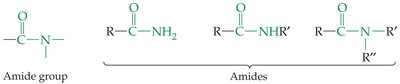
Amides have nitrogen bonded to a carbonyl carbon. They are important in biological molecules, such as proteins.
Heterocyclic Compounds
Structure and Significance
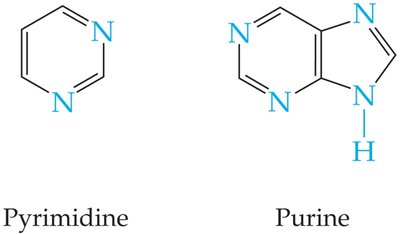
Heterocyclic compounds are rings containing atoms other than carbon, such as nitrogen, oxygen, or sulfur. Many are biologically important.
Alkaloids
Definition and Examples
Alkaloids are naturally occurring amines found in plants, bacteria, fungi, and animals. Many have physiological effects, including morphine, caffeine, nicotine, and cocaine. Pyrimidine and purine are also alkaloids.