 Back
BackOxidation and Reduction: Key Concepts and Applications
Study Guide - Smart Notes

Oxidation and Reduction
Introduction to Redox Reactions
Oxidation and reduction, collectively known as redox reactions, are fundamental chemical processes where electrons are transferred between substances. These reactions are essential in both industrial and biological contexts. - Oxidation: Loss of electrons, gain of oxygen, or loss of hydrogen. - Reduction: Gain of electrons, loss of oxygen, or gain of hydrogen. - Oxidation and reduction always occur together; when one substance is oxidized, another is reduced. 
Mnemonic for Redox Reactions
To remember the definitions: - "LEO the lion goes GER": LEO = Loss of Electrons is Oxidation; GER = Gain of Electrons is Reduction.
Characteristics of Oxidation and Reduction
The processes can be described in terms of oxygen, hydrogen, electrons, and oxidation numbers. 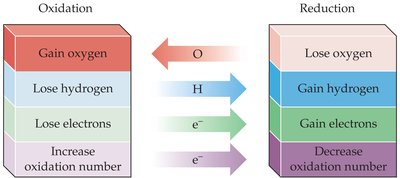 - Oxidation: Gain oxygen, lose hydrogen, lose electrons, increase oxidation number. - Reduction: Lose oxygen, gain hydrogen, gain electrons, decrease oxidation number.
- Oxidation: Gain oxygen, lose hydrogen, lose electrons, increase oxidation number. - Reduction: Lose oxygen, gain hydrogen, gain electrons, decrease oxidation number. 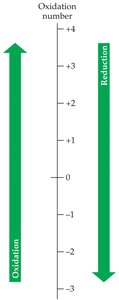
Examples of Redox Processes
Gain or Loss of Oxygen: - When a substance gains oxygen, it is oxidized. - When a substance loses oxygen, it is reduced. Gain or Loss of Hydrogen: - Loss of hydrogen is oxidation. - Gain of hydrogen is reduction. Gain or Loss of Electrons: - Loss of electrons is oxidation. - Gain of electrons is reduction.
Oxidizing and Reducing Agents
- Oxidizing agent: Causes oxidation by accepting electrons. - Reducing agent: Causes reduction by donating electrons.
Applications of Redox Reactions
Photochromic Glass
Photochromic lenses contain silver and copper(I) chloride. In sunlight, silver ions are reduced to silver atoms, darkening the lenses. - Reduction of silver ions: 

Electrochemical Cells and Batteries
Redox reactions can generate electricity in electrochemical cells. - Electrodes: Metal pieces where electron transfer occurs. - Anode: Site of oxidation. - Cathode: Site of reduction. 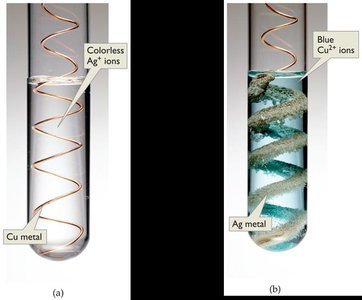
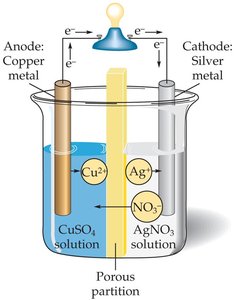 Half-reactions: - Oxidation: - Reduction: - Overall:
Half-reactions: - Oxidation: - Reduction: - Overall:
Types of Batteries
- Dry cell: Uses zinc and manganese dioxide. 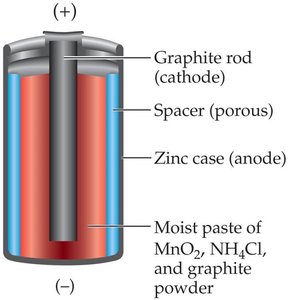 - Lead storage battery: Used in cars; can be recharged.
- Lead storage battery: Used in cars; can be recharged. 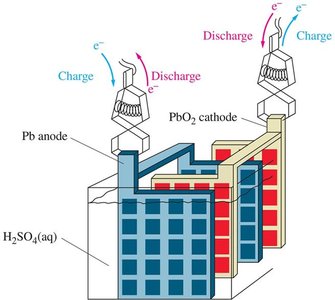
Corrosion and Protection
Rusting of Iron: Iron reacts with oxygen and water to form rust. 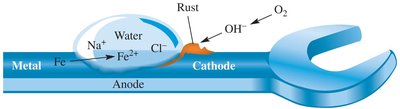 - Protection of Aluminum: Aluminum forms a tough oxide layer, preventing further corrosion. - Silver Tarnish: Silver reacts with hydrogen sulfide to form silver sulfide. Tarnish can be removed by reduction using aluminum and sodium bicarbonate.
- Protection of Aluminum: Aluminum forms a tough oxide layer, preventing further corrosion. - Silver Tarnish: Silver reacts with hydrogen sulfide to form silver sulfide. Tarnish can be removed by reduction using aluminum and sodium bicarbonate. 
Explosive Redox Reactions
Rapid redox reactions producing gases can cause explosions, such as the reaction of ammonium nitrate and fuel oil.
Common Oxidizing Agents
- Oxygen: Most abundant oxidizing agent; forms oxides with metals and nonmetals.  - Hydrogen peroxide: Used as disinfectant and bleach.
- Hydrogen peroxide: Used as disinfectant and bleach.  - Potassium dichromate: Used in breathalyzers; oxidizes alcohols. - Benzyl peroxide: Treats acne. - Chlorine: Disinfects water. - Bleaches: Sodium hypochlorite and calcium hypochlorite.
- Potassium dichromate: Used in breathalyzers; oxidizes alcohols. - Benzyl peroxide: Treats acne. - Chlorine: Disinfects water. - Bleaches: Sodium hypochlorite and calcium hypochlorite.
Common Reducing Agents
- Coke (carbon): Used in metallurgy to reduce metal oxides. - Aluminum: Reduces chromium oxide. - Hydroquinone: Reduces silver ions in photography.  - Antioxidants: Ascorbic acid (vitamin C), tocopherol (vitamin E), and vitamin A protect food from oxidation. - Hydrogen: Reduces metal oxides and unsaturated organic compounds. Catalysts like nickel or platinum speed up these reactions.
- Antioxidants: Ascorbic acid (vitamin C), tocopherol (vitamin E), and vitamin A protect food from oxidation. - Hydrogen: Reduces metal oxides and unsaturated organic compounds. Catalysts like nickel or platinum speed up these reactions.
Redox Reactions in Living Things
Biological Importance
Redox reactions are vital for life, providing energy through the oxidation of food. - Cellular respiration: - Photosynthesis:  Photosynthesis is the only process that produces elemental oxygen essential for animals. Example: Plants convert carbon dioxide and water into glucose and oxygen using sunlight. Additional info: Redox reactions are also involved in metabolism, detoxification, and cellular signaling.
Photosynthesis is the only process that produces elemental oxygen essential for animals. Example: Plants convert carbon dioxide and water into glucose and oxygen using sunlight. Additional info: Redox reactions are also involved in metabolism, detoxification, and cellular signaling.
