 Back
BackPeriodic Trends and Electron Configurations: Study Guide for Introductory Chemistry
Study Guide - Smart Notes

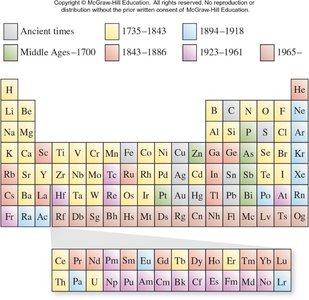
Periodic Trends of the Elements
Development of the Periodic Table
The periodic table is a fundamental tool in chemistry, organizing elements according to their atomic number and properties. Its development began with early observations of recurring chemical properties and culminated in the modern arrangement based on atomic number.
Law of Octaves: John Newlands observed that every eighth element had similar properties when arranged by atomic mass.
Mendeleev's Periodicity: Dmitri Mendeleev grouped elements by increasing atomic mass and predicted properties of undiscovered elements.
Moseley's Contribution: Henry Moseley established the importance of atomic number, resolving inconsistencies in the table.
Modern Table: Elements are arranged by atomic number and electron configuration.
Groups and Periods: Elements in the same group (vertical column) share similar properties.
Example: Elements in Group 17 (halogens) such as fluorine, bromine, and iodine exhibit properties similar to chlorine.
Electron Configurations
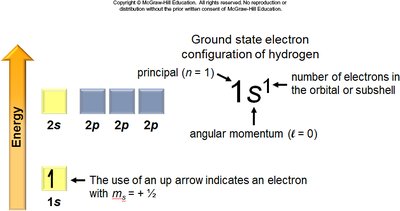
Electron configuration describes the distribution of electrons in atomic orbitals. The arrangement follows specific rules and principles, which determine chemical properties and reactivity.
Ground State: The lowest energy configuration for an atom's electrons.
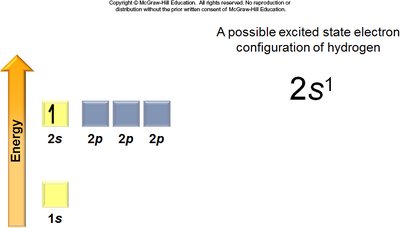
Excited State: Electrons occupy higher energy orbitals than in the ground state.
Quantum Numbers: Describe the properties of atomic orbitals and electrons (principal n, angular momentum l, magnetic ml, spin ms).
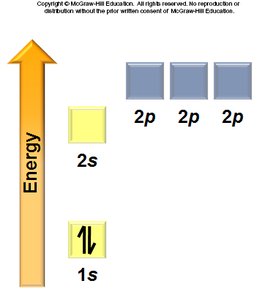
Pauli Exclusion Principle: No two electrons in an atom can have the same four quantum numbers.
Aufbau Principle: Electrons fill the lowest energy orbitals first.
Hund’s Rule: Electrons occupy degenerate orbitals singly before pairing.
General Rules: Orbitals fill in a specific order, each can hold two electrons, and electrons avoid pairing in degenerate orbitals if possible.
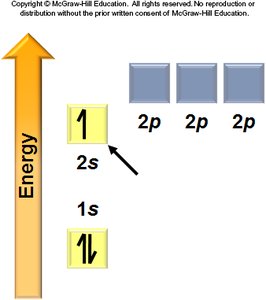
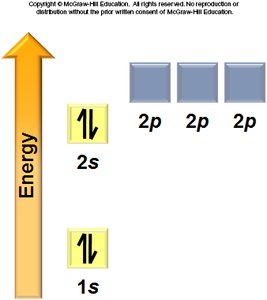
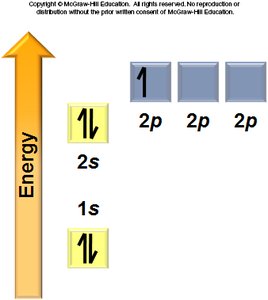
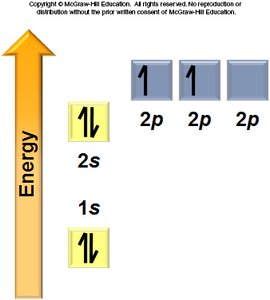
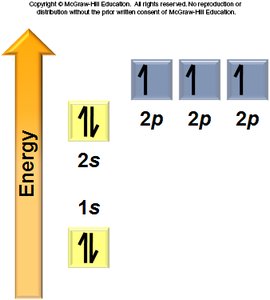
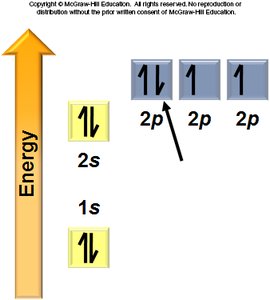
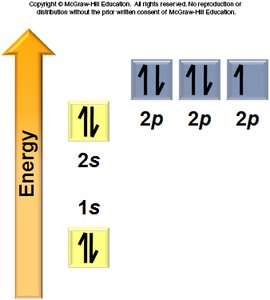
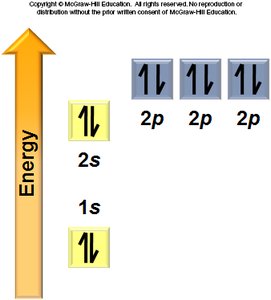
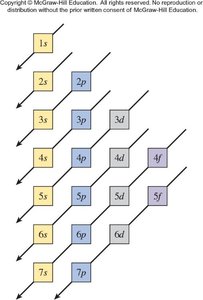
Electron Configurations and the Periodic Table
The periodic table reflects the electron configurations of elements, with similarities in valence electron arrangements leading to similar chemical properties.
Noble Gas Core: Electron configurations can be abbreviated using the previous noble gas.
Transition Metals: Groups 3–11 are transition metals, with unique electron filling patterns.
Lanthanides and Actinides: f-block elements with distinct electron configurations.
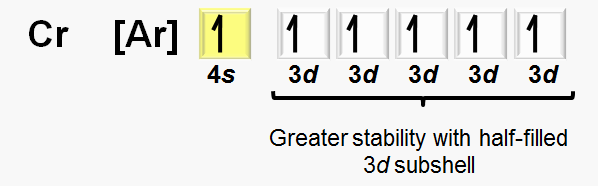
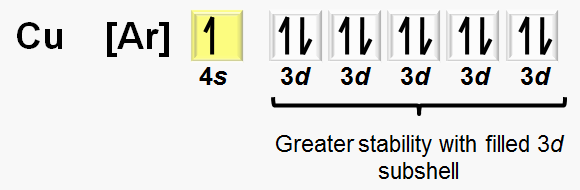
Exceptions: Some transition metals (e.g., chromium, copper) have anomalous electron configurations due to stability of half-filled or fully filled d subshells.
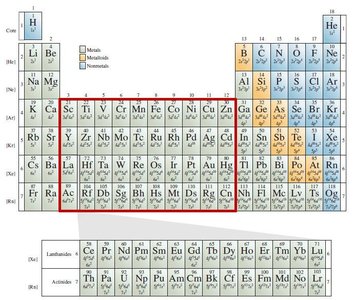
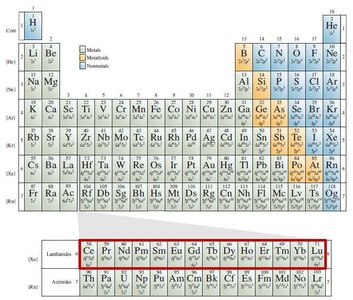
Classification of Elements
Elements are classified as main group elements, transition metals, noble gases, and f-block elements based on their electron configurations and periodic table location.
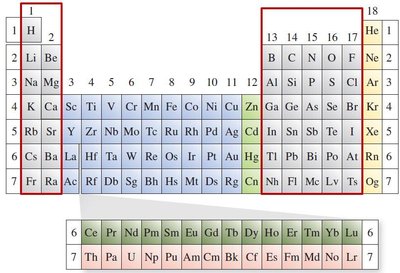
Main Group Elements: Groups 1, 2, and 13–17.
Noble Gases: Group 18, with completely filled p subshells.
Transition Metals: Groups 3–11, with partially filled d subshells.
Lanthanides and Actinides: f-block elements.
Valence Electrons and Chemical Properties
Valence electrons are the outermost electrons of an atom and are crucial in determining chemical reactivity and bonding.
Valence Configuration: Similarity in valence electron configuration predicts similar chemical properties.
Example: Group 1A elements have a single valence electron in an ns orbital.
Effective Nuclear Charge (Zeff)
Effective nuclear charge is the net positive charge experienced by an electron in a multi-electron atom, accounting for both attraction to the nucleus and repulsion by other electrons.
Shielding: Core electrons shield valence electrons from the full nuclear charge.
Trend: Zeff increases from left to right across a period, but changes little down a group.
Formula: where is the atomic number and is the shielding constant.
Periodic Trends in Properties of Elements
Atomic Radius
Atomic radius is the distance from the nucleus to the outermost electron shell. It varies depending on whether the element is a metal or nonmetal.
Metallic Radius: Half the distance between nuclei of adjacent metal atoms.
Covalent Radius: Half the distance between nuclei of bonded nonmetal atoms.
Trend: Increases down a group (higher n), decreases across a period (higher Zeff).
Ionization Energy
Ionization energy is the minimum energy required to remove an electron from an atom in the gas phase, forming a cation.
First Ionization Energy (IE1): Energy to remove the most loosely held electron.
Trend: Increases across a period (higher Zeff), decreases down a group.
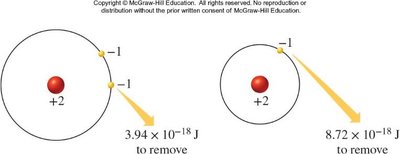
Successive Ionization Energies: Each additional electron removed requires more energy, especially when removing core electrons.
Electron Affinity
Electron affinity is the energy released when an atom in the gas phase accepts an electron, forming an anion.
Trend: Increases across a period (higher Zeff), but trends are less regular than for atomic radius or ionization energy.
Positive EA: Indicates a favorable process for electron addition.
Subsequent EA: Adding more than one electron is always less favorable due to repulsion.
Metallic Character
Metallic character describes the physical and chemical properties typical of metals, nonmetals, and metalloids.
Metals: Shiny, malleable, ductile, good conductors, low ionization energies.
Nonmetals: Varied color, brittle, poor conductors, high electron affinities.
Metalloids: Intermediate properties between metals and nonmetals.
Electron Configuration of Ions
To determine the electron configuration of ions, add electrons for anions and remove electrons for cations, following the rules for orbital filling.
Main Group Ions: Often become isoelectronic with the nearest noble gas.
d-Block Ions: Electrons are removed first from the highest n shell (usually s before d).
Isoelectronic Series: Ions with identical electron configurations but different nuclear charges; size decreases with increasing nuclear charge.
Ionic Radius
The ionic radius is the radius of a cation or anion. Cations are smaller than their parent atoms, while anions are larger. Ionic radius increases down a group and decreases with increasing charge in an isoelectronic series.
Isoelectronic Series: Size order: 3- > 2- > 1- > 1+ > 2+ > 3+
Chapter Summary: Key Points
Development of the Periodic Table
Electron Configurations and Rules
Classification of Elements
Valence Electron Configuration
Effective Nuclear Charge
Atomic Radius, Ionization Energy, Electron Affinity
Metallic Character
Ions and Isoelectronic Series
