 Back
BackQuantities in Chemical Reactions: Stoichiometry, Limiting Reactants, and Enthalpy
Study Guide - Smart Notes

Quantities in Chemical Reactions
Introduction to Reaction Stoichiometry
Stoichiometry is the quantitative study of reactants and products in a chemical reaction. It allows chemists to predict the amounts of substances consumed and produced in a given reaction, based on the balanced chemical equation.
Balanced chemical equations provide the numerical relationships between reactants and products.
These relationships are essential for calculating the quantities of materials required or produced.
Stoichiometry is foundational for laboratory work, industrial processes, and environmental chemistry.
Global Warming and Combustion of Fossil Fuels
Combustion Reactions and CO2 Production
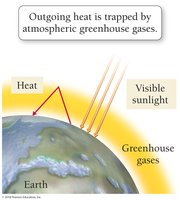
The combustion of fossil fuels, such as octane (a component of gasoline), produces water and carbon dioxide. Carbon dioxide is a greenhouse gas that contributes to global warming by trapping heat in the Earth's atmosphere.
The greenhouse effect occurs when greenhouse gases allow visible light to enter the atmosphere but prevent heat from escaping.
The balanced equation for octane combustion:
This equation shows that 16 moles of CO2 are produced for every 2 moles of octane burned.

Stoichiometry: Relationships Between Ingredients
Analogies with Recipes
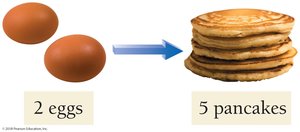
Stoichiometry can be compared to following a recipe, where the ingredients must be combined in specific ratios to produce a desired outcome. For example, making pancakes requires a certain ratio of flour, eggs, and baking powder.
Recipe: 1 cup flour + 2 eggs + 1/2 tsp baking powder → 5 pancakes
If you have 2 eggs and enough of everything else, you can make 5 pancakes.
The ratio is 2 eggs : 5 pancakes.


Mole-to-Mole Conversions in Chemical Equations
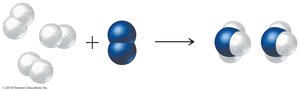
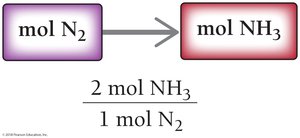
Balanced chemical equations act as recipes for chemical reactions, showing the ratios in which reactants combine and products form. For example, the synthesis of ammonia:
Equation:
Ratio: 3 mol H2 : 1 mol N2 : 2 mol NH3
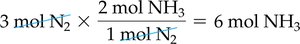
Mass-to-Mass Conversions
Using Molar Mass and Stoichiometric Ratios
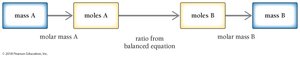
Often, chemists need to relate the mass of a reactant to the mass of a product. This requires converting grams to moles, using the balanced equation to relate moles of reactant to moles of product, and then converting back to grams.
General solution map:
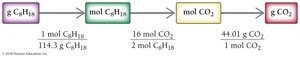
Example: What mass of CO2 is produced from 5.0 × 102 g of octane?
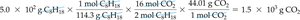
Limiting Reactant, Theoretical Yield, and Percent Yield
Limiting Reactant Concept
In a chemical reaction, the limiting reactant is the substance that is completely consumed first, thus limiting the amount of product formed. The theoretical yield is the maximum amount of product that can be formed from the limiting reactant.
Example (pancake analogy): Given 3 cups flour, 10 eggs, and 4 tsp baking powder, the limiting reactant is flour, allowing for 15 pancakes (theoretical yield).



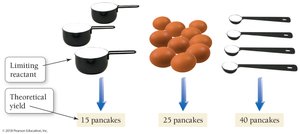
Actual Yield and Percent Yield
The actual yield is the amount of product actually obtained from a reaction, which is often less than the theoretical yield due to losses or side reactions. Percent yield is calculated as:
Example: If 11 pancakes are made out of a theoretical 15, percent yield is 73%.

Limiting Reactant and Percent Yield: Chemical Examples
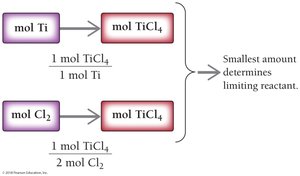
To determine the limiting reactant and theoretical yield in chemical reactions, use mole or mass relationships from the balanced equation. The reactant that produces the least amount of product is the limiting reactant.
Example: Ti(s) + 2 Cl2(g) → TiCl4(s)
Given: 1.8 mol Ti and 3.2 mol Cl2

Limiting Reactant and Percent Yield: Gram to Gram
When reactant quantities are given in grams, convert to moles, use stoichiometry to find the limiting reactant, and then calculate theoretical and percent yields.
Example: Na(s) + Cl2(g) → 2 NaCl(s)
Given: 53.2 g Na and 65.8 g Cl2
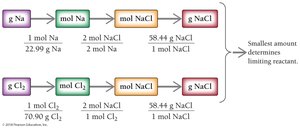
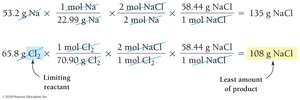
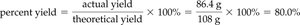
Limiting Reactant and Percent Yield: Additional Example
Example: Cu2O(s) + C(s) → 2 Cu(s) + CO(g)
Given: 11.5 g Cu2O and 114.5 g C
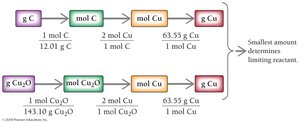


Enthalpy: Heat in Chemical Reactions
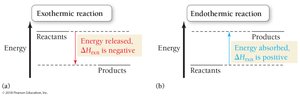
Definition and Sign of Enthalpy Change (ΔHrxn)
Enthalpy (ΔHrxn) is the heat exchanged in a chemical reaction at constant pressure. The sign of ΔHrxn indicates whether the reaction is exothermic (releases heat, negative ΔH) or endothermic (absorbs heat, positive ΔH).
Exothermic: Heat flows out of the system (ΔHrxn < 0)
Endothermic: Heat flows into the system (ΔHrxn > 0)
Stoichiometry of ΔHrxn
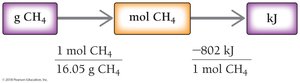
The magnitude of ΔHrxn is associated with the stoichiometric amounts of reactants and products in the balanced equation. It can be used as a conversion factor between moles and energy (kJ).
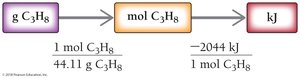
Example: Combustion of propane
1 mol C3H8 releases 2044 kJ of heat.
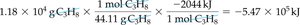
Everyday Chemistry: Bunsen Burners
Air Intake and Flame Characteristics
Bunsen burners allow adjustment of air intake, which affects the flame's color, temperature, and efficiency. Proper adjustment results in a hot, blue flame optimal for laboratory use.
No air: Yellow, smoky, cooler flame
Optimum air: Blue, non-smoky, hottest flame
Too much air: Cooler, may extinguish

Summary Table: Key Stoichiometric Concepts
Term | Definition |
|---|---|
Stoichiometry | Quantitative relationship between reactants and products in a chemical reaction |
Limiting Reactant | Reactant that is completely consumed first, limiting the amount of product formed |
Theoretical Yield | Maximum amount of product possible from the limiting reactant |
Actual Yield | Amount of product actually obtained from a reaction |
Percent Yield | (Actual yield / Theoretical yield) × 100% |
Enthalpy (ΔHrxn) | Heat released or absorbed by a reaction at constant pressure |
Learning Objectives
Recognize numerical relationships in balanced chemical equations.
Carry out mole-to-mole and mass-to-mass conversions.
Calculate limiting reactant, theoretical yield, and percent yield.
Calculate the amount of thermal energy emitted or absorbed in a reaction.
