 Back
BackLecture 15
Study Guide - Smart Notes

Chemical Reactions and Stoichiometry
Reactants, Products, and Balancing Equations
Chemical reactions involve the transformation of substances (reactants) into new substances (products). A balanced chemical equation shows the same number of each type of atom on both sides, reflecting the law of conservation of mass.
Reactants: Substances present before the reaction (e.g., CaO and H2O in CaO + H2O → Ca(OH)2).
Products: Substances formed as a result of the reaction (e.g., Ca(OH)2).
Balancing Equations: Adjust coefficients to ensure equal numbers of each atom on both sides.
Example: The equation CaO + H2O → Ca(OH)2 is already balanced (1 Ca, 2 H, 2 O on both sides).
Molecular, Molar, and Mass Relationships
Chemical equations can be interpreted in terms of molecules, moles, and mass. The coefficients indicate the ratio of molecules or moles, which can be converted to mass using molar masses.
Molecular Ratio: 1 molecule CaO reacts with 1 molecule H2O to form 1 molecule Ca(OH)2.
Molar Ratio: 1 mol CaO reacts with 1 mol H2O to form 1 mol Ca(OH)2.
Mass Ratio: Use molar masses to convert between grams and moles.

Practice: Mass Relationships in Chemical Reactions
To determine how much water is needed to produce a certain mass of Ca(OH)2:
Given: 2 kg (2000 g) Ca(OH)2
Molar mass Ca(OH)2: 74 g/mol
Molar mass H2O: 18 g/mol
Equation: CaO + H2O → Ca(OH)2
Steps:
Calculate moles of Ca(OH)2:
From the equation, 1 mol H2O per 1 mol Ca(OH)2
Calculate mass of H2O:
Solution Chemistry
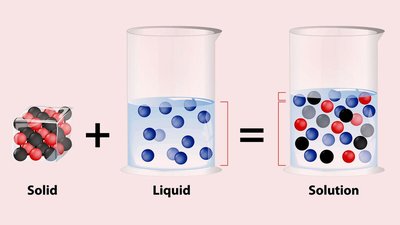
What is a Solution?
A solution is a homogeneous mixture of two or more substances. The solvent is the component present in the largest amount and does the dissolving, while the solute is the substance being dissolved.
Solvent: Major component (e.g., water in saltwater)
Solute: Minor component(s) (e.g., salt in saltwater)
Solution Concentration
The concentration of a solution describes the amount of solute in a given amount of solvent or solution. Solutions can be classified as dilute (small amount of solute) or concentrated (large amount of solute).
Molarity (M)
Molarity is a common unit of concentration, defined as moles of solute per liter of solution:
Example: To prepare 0.750 L of 0.15 M NaCl solution, calculate the required mass of NaCl:
Find moles:
Molar mass NaCl = 58.44 g/mol
Mass needed:
Percent Concentration
Percent concentration expresses the amount of solute as a percentage of the total solution, either by volume or mass.
Percent by Volume:
Percent by Mass:
Example (Volume): A pint of beer (500 mL) with 25 mL alcohol: ABV.
Example (Mass): Hummingbird food with 20 g sugar and 80 g water: sugar by mass.
Parts per Notation: Percent, Per Mille, ppm, ppb
Very dilute concentrations are often expressed as parts per hundred (percent), per thousand (per mille), per million (ppm), or per billion (ppb):
Unit | Definition | Decimal Equivalent |
|---|---|---|
Percent (%) | Parts per hundred | 3% = 0.03 |
Per mille (‰) | Parts per thousand | 3‰ = 0.003 |
ppm | Parts per million | 3 ppm = 0.000003 |
ppb | Parts per billion | 3 ppb = 0.000000003 |
These can be based on mass, volume, or a mixture (e.g., 1 mg/L = 1 ppm in water).
States of Matter and Intermolecular Forces
States of Matter
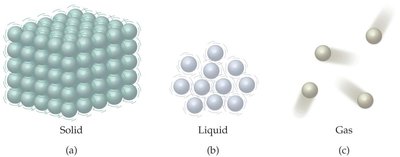
Matter exists in three primary states: solid, liquid, and gas. The arrangement and movement of particles differ in each state.
Solids: Particles have fixed positions and vibrate in place.
Liquids: Particles are close together but can move past each other.
Gases: Particles are far apart and move freely and rapidly.
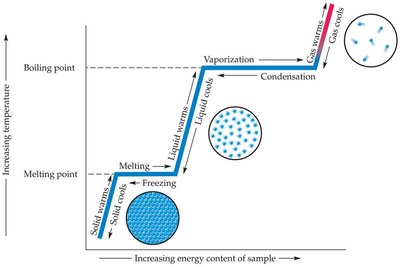
Changes of State
Transitions between states of matter are driven by energy changes:
Melting: Solid to liquid (at melting point)
Vaporization: Liquid to gas (at boiling point)
Condensation: Gas to liquid
Freezing: Liquid to solid (at freezing point)
Sublimation: Solid directly to gas
Intermolecular Forces
Intermolecular forces are attractions between molecules that determine the physical properties of substances, especially in liquids and solids. There are four main types:
Ionic Bonds: Attraction between cations and anions (strongest).
Dipole Forces: Attraction between polar molecules (includes ion-dipole forces).
Hydrogen Bonds: Special strong dipole force involving H bonded to N, O, or F.
Dispersion Forces: Temporary attractions due to momentary uneven electron distribution (weakest).
Ionic bonds are the strongest, while dispersion forces are the weakest. The type and strength of intermolecular forces affect melting and boiling points, solubility, and other properties.
Summary and Takeaways
Chemical equations represent the transformation of reactants to products and must be balanced.
Stoichiometry allows calculation of mass and mole relationships in reactions.
Solutions are homogeneous mixtures; concentration can be expressed in molarity, percent, ppm, etc.
Matter exists as solids, liquids, or gases, with changes of state driven by energy.
Intermolecular forces determine many physical properties of substances.
