 Back
BackSolutions and Their Properties: Structure, Solubility, and Colligative Effects
Study Guide - Smart Notes

Solutions and Their Properties
Introduction to Solutions
Solutions are homogeneous mixtures composed of two or more substances. The substance present in the largest amount is called the solvent, while the substance being dissolved is the solute. Solutions can exist in all three physical states: solid, liquid, and gas. In introductory chemistry, the focus is primarily on aqueous solutions, where water acts as the solvent.
The Dissolution Process
Formation of Solutions
The process of dissolving involves overcoming intermolecular forces (IMFs) between solute particles, solvent particles, and forming new solute-solvent interactions. The steps are:
Step 1: Overcome solute-solute IMFs (endothermic)
Step 2: Overcome solvent-solvent IMFs (endothermic)
Step 3: Form solute-solvent IMFs (exothermic)
The overall energy change determines if the process is exothermic, endothermic, or does not occur. The "like dissolves like" rule states that substances with similar IMFs are likely to be soluble in one another (e.g., polar solvents dissolve polar solutes).
Electrolytes and Nonelectrolytes
Definitions and Examples
Electrolytes: Substances that produce ions in solution and conduct electricity (e.g., NaCl, HCl).
Nonelectrolytes: Substances that do not produce ions in solution (e.g., sugar, ethanol).
Electrolytes are classified as strong (nearly 100% ionization) or weak (partial ionization). Ionic compounds dissociate physically, while some covalent compounds react chemically with water to form ions.
Solubility
Factors Affecting Solubility
Solubility is the maximum concentration of a solute that can dissolve in a solvent under specific conditions. Key factors include:
Nature of solute and solvent
Temperature
Pressure (for gases)
Terms:
Saturated solution: Contains the maximum amount of solute.
Unsaturated solution: Contains less than the maximum solute.
Supersaturated solution: Contains more than the maximum solute (unstable).
Temperature and Gas Solubility
The solubility of gases in water decreases as temperature increases. This is important for environmental and biological systems.
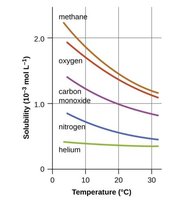
Pressure and Gas Solubility: Henry's Law
Gas solubility is directly proportional to the partial pressure of the gas above the solution. This relationship is described by Henry's Law:
Where is the concentration of the gas, is a constant, and is the partial pressure of the gas.

Example: Opening a carbonated beverage reduces the pressure above the liquid, causing dissolved CO2 to escape as bubbles.
Environmental Impact Example
Large-scale releases of dissolved gases, such as the 1986 Lake Nyos disaster, can have catastrophic effects on local populations and ecosystems.

Solubility of Liquids in Liquids
Miscible: Liquids that mix in all proportions (e.g., water and ethanol).
Immiscible: Liquids that do not mix appreciably (e.g., oil and water).
Partially miscible: Liquids with moderate mutual solubility (e.g., bromine and water).

Temperature and Solubility of Solids
The solubility of most solids increases with temperature. This is because dissolving a solid is usually an endothermic process.
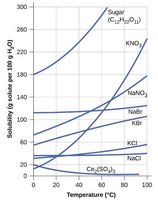
Colligative Properties
Definition and Types
Colligative properties depend on the number of solute particles, not their identity. Major colligative properties include:
Vapor pressure lowering
Boiling point elevation
Freezing point depression
Osmotic pressure
Concentration Units
Molarity (M): Moles of solute per liter of solution (temperature dependent).
Molality (m): Moles of solute per kilogram of solvent (temperature independent).
Mole fraction (X): Moles of component divided by total moles.
Vapor Pressure Lowering and Raoult's Law
Adding a nonvolatile solute lowers the vapor pressure of the solvent. Raoult's Law describes this effect:
Where is the vapor pressure of the solution, is the mole fraction of the solvent, and is the vapor pressure of the pure solvent.
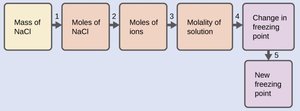
Boiling Point Elevation and Freezing Point Depression
Both properties are a direct result of vapor pressure lowering. The equations are:
Where and are the boiling point elevation and freezing point depression constants, respectively, and is the molality of the solution.
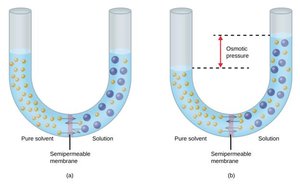
Osmosis and Osmotic Pressure
Osmosis is the net movement of solvent molecules through a semipermeable membrane from a region of lower solute concentration to higher solute concentration. The osmotic pressure () is given by:
Where is molarity, is the gas constant, and is temperature in Kelvin.
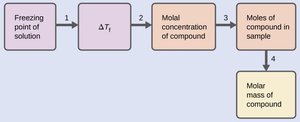
Determination of Molar Mass from Colligative Properties
Colligative properties can be used to determine the molar mass of an unknown solute by measuring the change in freezing point or osmotic pressure.
Colligative Properties of Electrolytes and the van't Hoff Factor
Electrolytes produce more particles in solution than nonelectrolytes, increasing the effect on colligative properties. The van't Hoff factor () is the ratio of actual particles in solution to formula units dissolved. For NaCl, is approximately 2; for glucose, is 1. In practice, may be less than predicted due to ion pairing.
Colloids
Definition and Properties
Colloids are mixtures where the dispersed particles are larger than those in solutions but do not settle out. Examples include milk, fog, and gelatin. Colloids scatter light (Tyndall effect) and have important technological and biological applications.

Table: Comparison of Mixtures
Type | Particle Size | Appearance | Settling |
|---|---|---|---|
Solution | <1 nm | Clear | No |
Colloid | 1–1000 nm | Cloudy | No |
Suspension | >1000 nm | Cloudy | Yes |
Additional info: Colloids are stabilized by electrostatic or steric repulsion, and their unique properties are exploited in food, medicine, and environmental technology.
