 Back
BackLecture 16
Study Guide - Smart Notes

States of Matter
Classification of Matter
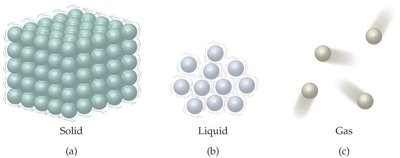
The physical state of matter is determined by the arrangement and movement of its particles. The three primary states are solid, liquid, and gas. Each state has distinct properties based on particle arrangement and energy.
Solids: Particles are closely packed in a fixed structure, resulting in definite shape and volume.
Liquids: Particles are close but can move past each other, giving liquids a definite volume but no fixed shape.
Gases: Particles are far apart and move freely, resulting in neither definite shape nor volume.
Example: Water exists as ice (solid), liquid water, and water vapor (gas).
Changes of State
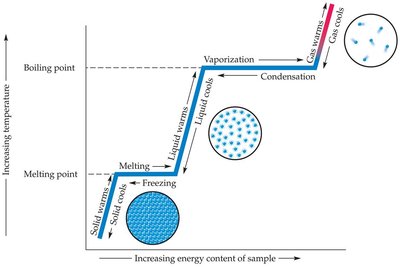
Transitions between states of matter are driven by energy changes. Key processes include:
Melting: Solid to liquid at the melting point.
Freezing: Liquid to solid at the freezing point (same temperature as melting point).
Vaporization: Liquid to gas at the boiling point.
Condensation: Gas to liquid.
Sublimation: Solid directly to gas.
Example: Ice melting to water, water boiling to steam.
Intermolecular Forces
Overview of Intermolecular Forces
Intermolecular forces are attractions between molecules that determine the physical properties of substances, such as boiling and melting points. There are four main types, ranked by strength:
Ionic Bonds: Attraction between cations and anions; strongest force in condensed states.
Dipole Forces: Attraction between polar molecules.
Hydrogen Bonds: Special dipole force involving hydrogen bonded to N, O, or F.
Dispersion Forces: Temporary attractions due to induced dipoles; weakest force.
Ionic Bonds
Ionic bonds occur between ions of opposite charge, such as Na+ and Cl-. These bonds are responsible for the structure of ionic solids and are the strongest intermolecular force.
Example: Sodium chloride (NaCl) forms a crystalline solid due to ionic bonding.
Dipole Forces
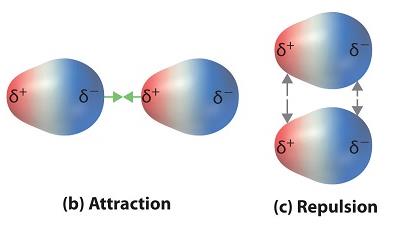
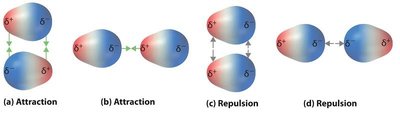
Dipole forces arise in polar molecules, which have regions of partial positive and negative charge. These molecules attract each other through their dipoles.
Attraction: Opposite charges attract.
Repulsion: Like charges repel, but attractive forces dominate in liquids.
Example: Water (H2O) molecules attract each other via dipole forces.
Ion-Dipole Forces
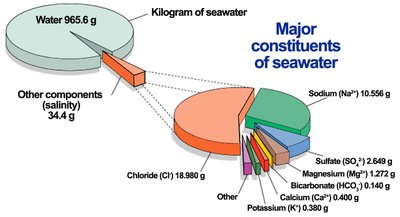
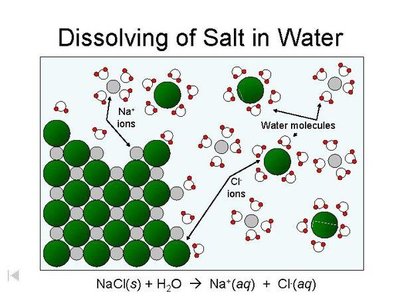
Ion-dipole forces occur when ions interact with polar molecules, especially in solutions. These forces are crucial for dissolving ionic compounds in water.
Example: Na+ and Cl- ions are surrounded by water molecules in seawater.
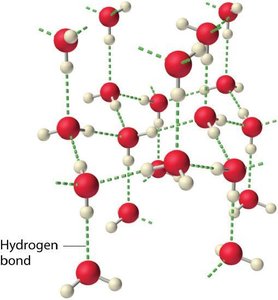
Hydrogen Bonds
Hydrogen bonds are a special type of dipole force. They occur when hydrogen is covalently bonded to highly electronegative atoms (N, O, F), resulting in strong attractions between molecules.
Example: Water exhibits strong hydrogen bonding, leading to high boiling point and unique properties of ice.
Ice Structure: Hydrogen bonds create an open, cage-like structure, making ice less dense than liquid water.
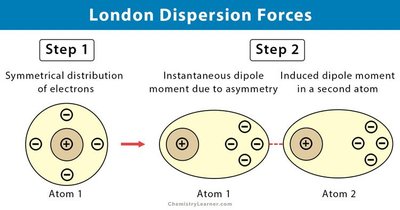
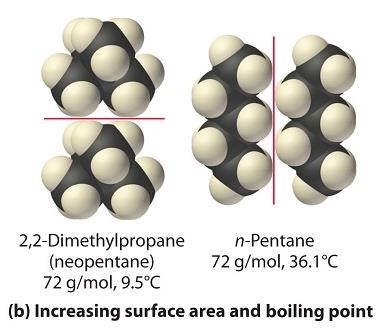
Dispersion Forces (London Dispersion Forces)
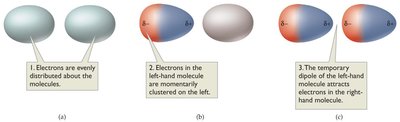
Dispersion forces are present in all molecules, but are the only intermolecular force in nonpolar molecules. They arise from temporary dipoles induced by electron movement.
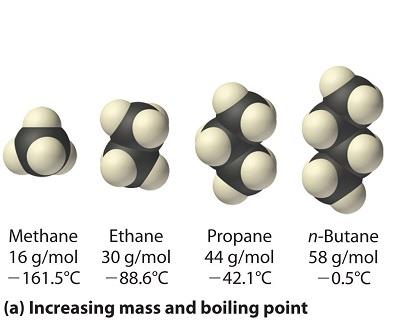
Strength: Increases with molecular weight and surface area.
Example: Hydrocarbons like methane, ethane, propane, and butane show increasing boiling points with mass.
Intermolecular Forces and Solutions
Solubility and "Like Dissolves Like"
Solubility depends on the similarity of intermolecular forces between solute and solvent. Polar solvents dissolve polar solutes, and nonpolar solvents dissolve nonpolar solutes.
Example: Water (polar) dissolves salt (ionic), but not oil (nonpolar).
Summary Table: Types of Intermolecular Forces
Type | Strength | Example | Key Feature |
|---|---|---|---|
Ionic Bonds | Strongest | NaCl | Cation-anion attraction |
Dipole Forces | Strong | H2O | Polar molecule attraction |
Hydrogen Bonds | Moderate-Strong | H2O, NH3 | H bonded to N, O, or F |
Dispersion Forces | Weakest | CH4, C6H6 | Temporary dipoles |
Key Takeaways
States of matter are determined by particle arrangement and energy.
Intermolecular forces govern physical properties and solubility.
Four main types of intermolecular forces: ionic, dipole, hydrogen, and dispersion.
"Like dissolves like" is a guiding principle for solution formation.
