 Back
BackStudy Guide: Chemical Reactions and Solutions in Introductory Chemistry
Study Guide - Smart Notes

The Mole and Avogadro's Number
Definition and Importance
The concept of the mole is fundamental in chemistry, allowing scientists to count atoms, molecules, or ions in a given sample using a standard quantity.
Mole: A unit representing 6.022142 x 1023 particles, known as Avogadro's Number.
Avogadro's Number: The number of particles in exactly 12 grams of carbon-12 (12C).
Allows chemists to relate microscopic particles to macroscopic amounts.

Balancing Chemical Equations
Principles and Practice
Chemical equations must be balanced to obey the law of conservation of mass, ensuring the same number of atoms for each element on both sides of the equation.
Use coefficients to balance atoms.
Start with elements that appear only once on each side.
Balance hydrogen and oxygen last.
Example: Ammonia reacts with oxygen to yield nitrogen monoxide and water.
Balanced Equation: 
Example: Aluminum Sulfide and Water
Aluminum sulfide reacts with water to yield aluminum hydroxide and dihydrogen sulfide.
Balanced Equation:

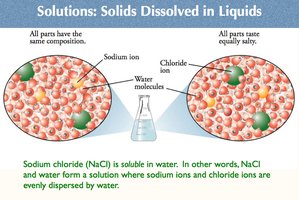
Solutions: Solids Dissolved in Liquids
Formation and Properties
Solutions are homogeneous mixtures where a solute is dissolved in a solvent.
Solute: The minor component, dispersed in the solvent.
Solvent: The major component, disperses the solute.
Example: Sodium chloride (NaCl) dissolves in water, forming a solution with sodium and chloride ions evenly dispersed.
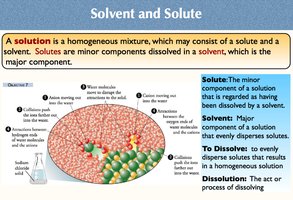
Solvent and Solute
Definitions and Interactions
A solution is a homogeneous mixture of solute and solvent.
Solute: Minor component, dissolved by the solvent.
Solvent: Major component, disperses the solute.
Dissolution: The process of dissolving.
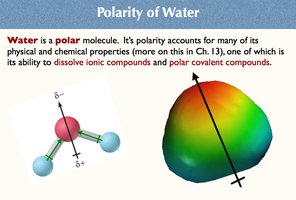
Polarity of Water
Role in Dissolving Compounds
Water is a polar molecule, meaning it has a partial positive charge on one end and a partial negative charge on the other.
This polarity allows water to dissolve ionic and polar covalent compounds.
Polarity is responsible for many physical and chemical properties of water.
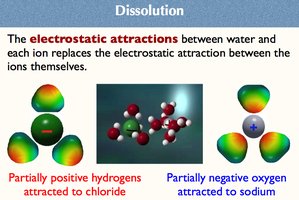
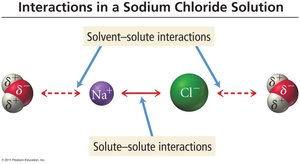
Dissolution and Solute-Solvent Interactions
Electrostatic Attractions
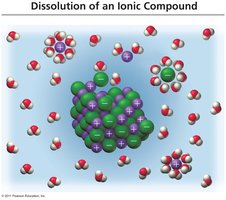
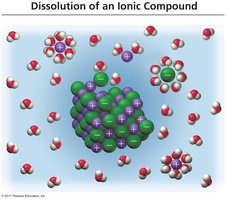
When ionic compounds dissolve, water molecules surround the ions, replacing the electrostatic attraction between the ions themselves.
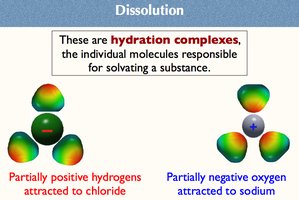
Partially positive hydrogens are attracted to chloride ions.
Partially negative oxygen atoms are attracted to sodium ions.
Hydration Complexes
Hydration complexes are formed when water molecules surround ions, solubilizing them.
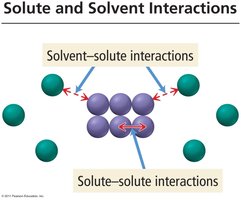
Solute and Solvent Interactions
Solute-solute interactions: Forces between solute particles.
Solvent-solvent interactions: Forces between solvent molecules.
Solvent-solute interactions: Forces between solute and solvent, crucial for dissolution.
Dissociation
Process and Examples
Dissociation is the process of separating cations and anions by water, resulting in free ions in solution.
Example:
Example:
The (aq) symbol indicates ions are solvated by water.

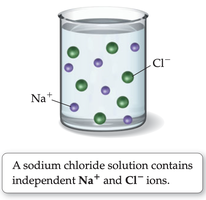
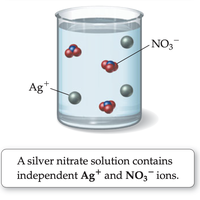
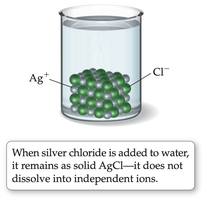

Salt vs. Sugar Dissolved in Water
Ionic vs. Molecular Compounds
Ionic compounds dissociate into ions when dissolved in water.
Molecular compounds do not dissociate; they remain intact.
Ionic Solutions Conduct Electricity
Electrolytes and Conductivity
Electrolyte: A substance that conducts electricity when dissolved in water.
Strong electrolytes dissociate completely and conduct electricity best.
Example: NaCl solution conducts electricity, AgCl does not.

Measure of Solubility
Saturation and Solubility Limits
Solubility is the maximum amount of solute that can dissolve in a solvent at a given temperature and pressure.
Saturation: The limit of solubility.
Solubility is reported as a concentration unit (e.g., g/L).
Example: Solubility of NaCl in water at 20°C is 365 g/L; AgCl is 0.009 g/L.

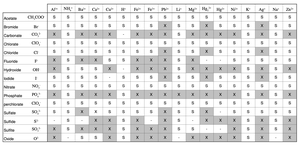
Solubility Table
Solubility tables classify compounds as soluble (S) or insoluble (X) with various ions.
Anion | Na+ | K+ | Ba2+ | Ca2+ | Fe2+ | Fe3+ | Li+ | Mg2+ | Ag+ | Zn2+ |
|---|---|---|---|---|---|---|---|---|---|---|
Acetate | S | S | S | S | S | S | S | S | S | S |
Bromide | S | S | S | S | S | S | S | S | X | S |
Chloride | S | S | S | S | S | S | S | S | X | S |
Sulfate | S | S | X | X | S | S | S | S | S | S |
Phosphate | X | X | X | X | X | X | X | X | X | X |
Carbonate | X | X | X | X | X | X | X | X | X | X |
Precipitation Reactions
Formation and Identification
Precipitation reactions occur when two solutions are mixed and an insoluble product (precipitate) forms.
Precipitate: A solid substance formed during a reaction that is insoluble in the solvent.
Electrostatic attractions between ions outweigh hydration, causing precipitation.
Example:

Summary Table: Key Concepts
Concept | Definition | Example |
|---|---|---|
Mole | 6.022 x 1023 particles | 1 mole of NaCl |
Solute | Minor component in solution | NaCl in water |
Solvent | Major component in solution | Water in NaCl solution |
Dissociation | Separation of ions by water | KBr(s) → K+(aq) + Br-(aq) |
Precipitate | Insoluble solid formed in reaction | PbI2(s) in NaI + Pb(NO3)2 |
Additional info:
Some images and tables were inferred to clarify solubility and dissociation concepts.
Solubility table entries are representative and may be abbreviated for clarity.
