 Back
BackUnderstanding Precision and Accuracy in Chemistry Measurements
Study Guide - Smart Notes

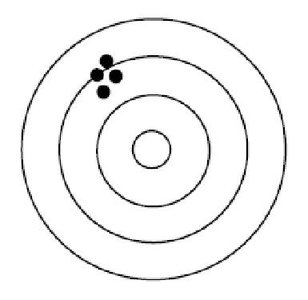
Q7. The figure below represents the bull's eye target for an archer. The black dots represent where the archer’s arrows hit. How can this archer be described?
Background
Topic: Precision and Accuracy in Measurements
This question tests your understanding of the difference between precision and accuracy, which are important concepts in scientific measurements and data analysis.
Key Terms:
Accuracy: How close a measured value is to the true or accepted value.
Precision: How close repeated measurements are to each other, regardless of whether they are close to the true value.
Step-by-Step Guidance
Examine the pattern of the black dots (arrows) on the target. Are they close together, or are they spread out?
Consider where the cluster of arrows is located relative to the center (bull's eye) of the target. Are they near the center, or off to one side?
Recall that high precision means the arrows are grouped closely together, while high accuracy means the arrows are close to the bull's eye.
Think about which description best fits the pattern shown: precise, accurate, both, or neither.
Try solving on your own before revealing the answer!
Final Answer: A) precise
The arrows are closely grouped together (high precision), but they are not centered on the bull's eye (not accurate). This means the archer is precise but not accurate.
