 Back
BackWater: Properties, Contamination, and Treatment (Introductory Chemistry Study Notes)
Study Guide - Smart Notes

Water: Some Unique Properties
Density and Expansion Upon Freezing
Water is a remarkable substance with several unique physical properties that are essential for life and environmental processes. One of its most notable characteristics is its relatively high density as a liquid and its unusual expansion upon freezing. Unlike most substances, water becomes less dense as it transitions from liquid to solid, which is why ice floats on water.
High Density (Liquid): Water molecules are closely packed due to hydrogen bonding, resulting in a high density compared to other liquids.
Expansion Upon Freezing: When water freezes, its molecules arrange in a crystalline structure that is less dense than liquid water, causing ice to float.
Environmental Impact: Floating ice insulates aquatic life in cold climates and influences global climate patterns.
Example: Lakes freeze from the top down, allowing organisms to survive beneath the ice layer.
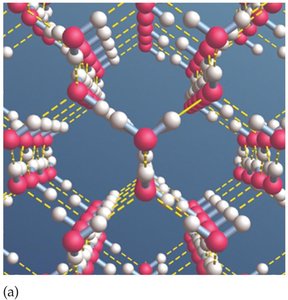

Hydrogen Bonding and Solvent Properties
Water's ability to form hydrogen bonds makes it an exceptional solvent, often referred to as the "universal solvent." This property allows water to dissolve a wide variety of substances, facilitating chemical reactions in biological and environmental systems.
Hydrogen Bonding: The attraction between the hydrogen atom of one water molecule and the oxygen atom of another creates a network of hydrogen bonds.
Universal Solvent: Water dissolves ionic compounds, polar molecules, and gases, making it essential for life and geochemical cycles.
Amphoteric Nature: Water can act as both an acid and a base, participating in acid-base reactions.
Example: Salt (NaCl) dissolves readily in water due to ion-dipole interactions.
High Heat of Vaporization
Water has a very high heat of vaporization, meaning it requires a large amount of energy to convert from liquid to gas. This property is crucial for regulating temperature in living organisms and the environment.
Definition: Heat of vaporization is the energy required to vaporize one gram of a substance at its boiling point.
Biological Importance: Evaporation of sweat cools the body efficiently due to water's high heat of vaporization.
Specific Heat Capacity
Specific heat is the amount of energy needed to raise the temperature of 1 gram of a substance by 1°C. Water's specific heat is much higher than most other substances, allowing it to moderate Earth's climate and stabilize temperatures in organisms.
Formula:
q: Heat absorbed or released (cal or J)
m: Mass (g)
C: Specific heat (cal/g°C or J/g·K)
ΔT: Change in temperature (°C or K)
Example Calculation: To raise 225 g of water from 25.0°C to 100.0°C:
To convert to joules:
Comparison Table: Water's specific heat is much higher than metals and other common substances.
Substance | Specific Heat (cal/g°C) | Specific Heat (J/g·K) |
|---|---|---|
Water (liquid) | 1.00 | 4.184 |
Aluminum (Al) | 0.216 | 0.902 |
Copper (Cu) | 0.0920 | 0.385 |
Ethanol (C2H5OH) | 0.588 | 2.46 |
Iron (Fe) | 0.107 | 0.449 |
Mercury (Hg) | 0.0332 | 0.139 |
Lead (Pb) | 0.0306 | 0.128 |
Water in Nature
Distribution of Water on Earth
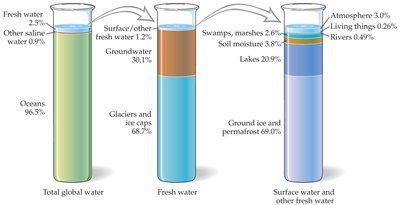
Water covers about 75% of Earth's surface, but the vast majority is seawater. Only a small fraction is available as fresh water for human use.
Seawater: Approximately 98% of Earth's water is found in oceans.
Freshwater: About 2% is locked in polar ice caps and glaciers, with only a tiny fraction available as liquid freshwater in lakes, rivers, and groundwater.
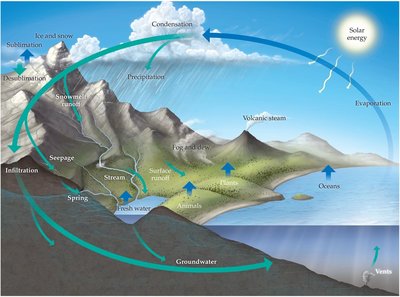
The Water Cycle
The water cycle describes the continuous movement of water on, above, and below the surface of the Earth. It is driven by solar energy and involves processes such as evaporation, condensation, precipitation, infiltration, and runoff.
Evaporation: Water changes from liquid to vapor, mainly from oceans and lakes.
Condensation: Water vapor cools and forms clouds.
Precipitation: Water returns to Earth's surface as rain, snow, or hail.
Infiltration and Runoff: Water seeps into the ground or flows over land into bodies of water.
The Water Cycle and Natural Contaminants
Natural Contaminants in Water
Natural waters contain a variety of dissolved substances and contaminants, both inorganic and organic. These can affect water quality and suitability for consumption.
Gases: Radon, carbon dioxide, nitrogen, and oxygen from the atmosphere.
Dissolved Minerals: Cations (e.g., calcium, magnesium, iron) and anions (e.g., chloride, sulfate, bicarbonate) from soil and rocks. Calcium and magnesium salts cause hard water.
Organic Matter: Bacteria, microorganisms, and animal wastes.
Substance | Formula | Source |
|---|---|---|
Carbon dioxide | CO2 | Atmosphere |
Nitrogen | N2 | Atmosphere |
Oxygen | O2 | Atmosphere |
Nitric acid | HNO3 | Atmosphere (thunderstorms) |
Calcium ions | Ca2+ | Limestone rocks |
Magnesium ions | Mg2+ | Dolomite rocks |
Iron(III) ions | Fe3+ | Soil and rocks |
Chloride ions | Cl− | Soil, rocks, fertilizer |
Sulfate ions | SO42− | Soil, rocks, fertilizer |
Bicarbonate ions | HCO3− | Soil and rocks |
Radon | Rn | Radioactive decay |
Chemical and Biological Contamination
Waterborne Disease
Contamination of water by pathogenic organisms is a major public health concern. Waterborne diseases can result from bacteria, viruses, and protozoa present in untreated or poorly treated water.
EPA Estimate: 30 million people in the U.S. are at risk from bacterial contamination.
Global Impact: Approximately 80% of illnesses worldwide are linked to water contamination.
Acid Rain
Acid rain is caused by the deposition of sulfur and nitrogen oxides (SOx and NOx) from industrial emissions. Acidic precipitation lowers the pH of soil and water bodies, causing environmental damage.
Effects: Damages vegetation, aquatic life, and infrastructure (e.g., corrosion of metals, dissolution of limestone and marble).
Sewage and Eutrophication
The release of sewage into water bodies increases the biochemical oxygen demand (BOD), leading to eutrophication—the accelerated aging of lakes due to nutrient enrichment. This process can result in oxygen depletion and loss of aquatic life.
Aerobic Decay: Organic matter decomposes in the presence of oxygen.
Anaerobic Decay: Decomposition occurs without oxygen, often producing foul-smelling gases.
Human Impact: Runoff from agriculture and urban areas accelerates eutrophication.
Groundwater Contamination
Sources and Risks
Groundwater is a major source of drinking water, but it is vulnerable to contamination from various sources. Once contaminated, groundwater is difficult and costly to clean.
Nitrates: Agricultural fertilizers and animal wastes contribute nitrates, which are highly soluble and difficult to remove. In infants, nitrates can cause methemoglobinemia (blue baby syndrome).
Volatile Organic Chemicals (VOCs): Industrial solvents, petroleum products, and landfill leachates can contaminate groundwater, posing health risks including cancer.
Making Water Fit to Drink
Water Use and Regulation
The United States has extensive water use and a large number of public water systems. The Safe Drinking Water Act (1974, amended 1986 and 1996) empowers the EPA to set and enforce standards for drinking water quality.
EPA Standards: Maximum contaminant levels (MCLs) are set for various inorganic and organic substances.
Substance | Maximum Contaminant Level (mg/L) |
|---|---|
Arsenic | 0.010 |
Barium | 2 |
Copper | 1.3 |
Lead | 0.015 |
Nitrate ion | 10 |
Atrazine (organic) | 0.003 |
Benzene (organic) | 0.005 |
Secondary Standards: Nonenforceable guidelines for taste, odor, and appearance (e.g., chloride, iron, sulfate).
Measuring Contaminants: ppm and ppb
Contaminant concentrations are often measured in parts per million (ppm) or parts per billion (ppb).
ppm:
ppb:
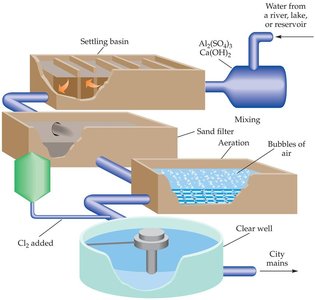
Water Treatment Plants
Coagulation and Filtration
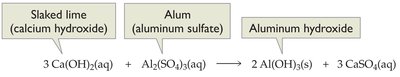
Urban water treatment involves several steps to remove contaminants and pathogens. The first step is coagulation, where slaked lime (calcium hydroxide) and alum (aluminum sulfate) are added to form gelatinous aluminum hydroxide, which traps colloidal particles and bacteria. The mixture is then filtered through sand and gravel.
Chemical Equation:
Chemical Disinfection
After filtration, chlorine is added to kill remaining bacteria and provide residual protection. Ozone and ultraviolet (UV) light can also be used for disinfection, with ozone being effective against viruses and UV suitable for small-scale applications.
Chlorination: Provides ongoing protection in the distribution system.
Ozonation: Effective against bacteria and viruses, but does not provide residual protection.
UV Light: Effective for small-scale disinfection, but no residual effect.
Fluoridation
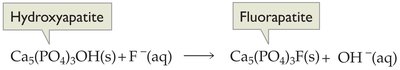
Fluoride is added to many municipal water supplies to prevent tooth decay. Fluoride ions replace hydroxide ions in tooth enamel, forming a harder, more acid-resistant mineral called fluorapatite.
Hydroxyapatite: (main component of tooth enamel)
Fluorapatite Formation:
Optimal Concentration: 0.7–1.0 ppm fluoride in drinking water.
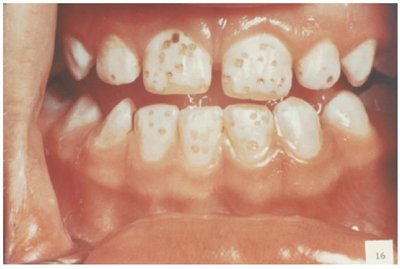
Risks: Excessive fluoride can cause dental fluorosis (discoloration of teeth).
Wastewater Treatment
Primary Treatment
Primary sewage treatment involves holding wastewater in settling ponds or tanks to allow heavier solids to precipitate as sludge. This step removes large particles and some organic matter.
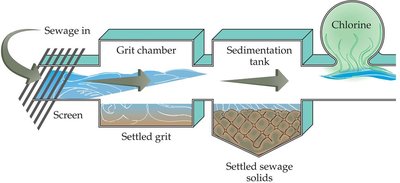
Secondary Treatment
Secondary treatment passes the effluent through sand and gravel filters, where aerobic bacteria break down much of the remaining organic matter. The activated sludge method uses aeration tanks to promote bacterial decomposition.
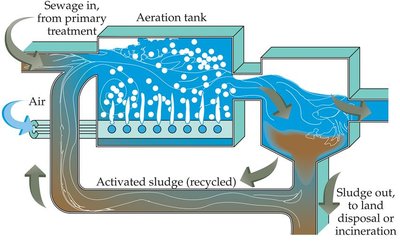
Tertiary Treatment and Alternatives
Tertiary treatment involves advanced processes such as charcoal filtration, reverse osmosis, and distillation to remove remaining contaminants. Alternative systems include the use of marshes for natural filtration and composting toilets that require no water or energy.
Sludge Use: Treated sludge can be used as fertilizer.
Natural Filtration: Marshes and wetlands can filter sewage and utilize nutrients.
