The principal short-term energy storage molecule in cells is __________ .
Ch. 2 - The Chemistry of Microbiology

Bauman6th EditionMicrobiology with Diseases by TaxonomyISBN: 9780134832302Not the one you use?Change textbook
Chapter 2, Problem 2
Name five properties of water that are vital to life.
 Verified step by step guidance
Verified step by step guidance1
Understand that water's unique properties arise from its molecular structure and hydrogen bonding, which are essential for supporting life processes.
Identify the first property: Cohesion and adhesion, which allow water molecules to stick to each other and to other surfaces, facilitating transport in plants and other organisms.
Recognize the second property: High specific heat capacity, meaning water can absorb or release a large amount of heat with little temperature change, helping to stabilize environmental and cellular temperatures.
Note the third property: High heat of vaporization, which enables cooling through evaporation, such as sweating in animals.
List the fourth and fifth properties: Water is a universal solvent, dissolving many substances necessary for biochemical reactions, and it has a solid form (ice) that is less dense than its liquid form, allowing ice to float and insulate aquatic life in cold environments.

Verified video answer for a similar problem:
This video solution was recommended by our tutors as helpful for the problem above.
Video duration:
5mWas this helpful?
Key Concepts
Here are the essential concepts you must grasp in order to answer the question correctly.
Cohesion and Adhesion
Cohesion refers to water molecules sticking to each other due to hydrogen bonding, enabling surface tension. Adhesion is water's ability to stick to other substances, aiding processes like capillary action in plants.
Recommended video:
Guided course
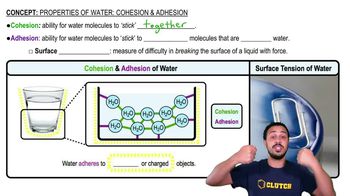
Properties of Water- Cohesion and Adhesion
High Specific Heat Capacity
Water can absorb or release large amounts of heat with minimal temperature change, helping organisms and environments maintain stable temperatures essential for life.
Recommended video:
Guided course

Water’s High Specific Heat
Solvent Properties
Water is a universal solvent because it dissolves many substances, facilitating chemical reactions and transport of nutrients and waste in biological systems.
Recommended video:
Guided course
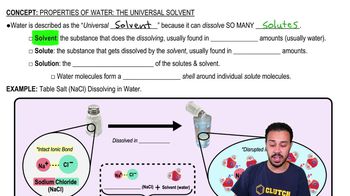
Properties of Water- The Universal Solvent
Related Practice
Textbook Question
793
views
Textbook Question
The atomic mass of an atom most closely approximates the sum of the masses of all its __________.
a. protons
b. isotopes
c. electrons
d protons and neutrons
757
views
Textbook Question
One isotope of iodine differs from another in __________ .
a. the number of protons
b. the number of electrons
c. the number of neutrons
d. atomic number
751
views
Textbook Question
The type of chemical bond between atoms with nearly equal electronegativities is called a(n) _____________ bond.
1561
views
Textbook Question
Describe the difference(s) among saturated fatty acids, unsaturated fatty acids, and polyunsaturated fatty acids.
1709
views
Textbook Question
Shown is the amino acid tryptophan. Put the letter “C” at the site of every carbon atom. Label the amino group, the carboxyl group, and the side group.
1480
views
