Textbook Question
The outermost electron shell of an atom is known as the _____________ shell.
863
views

 Verified step by step guidance
Verified step by step guidance

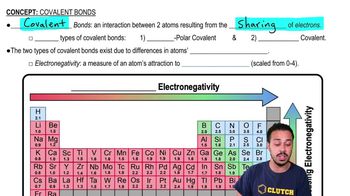

The outermost electron shell of an atom is known as the _____________ shell.
Which of the following structures have no electrical charge?
a. Protons
b. Electrons
c. Neutrons
d. Ions
The atomic mass of an atom most closely approximates the sum of the masses of all its __________.
a. protons
b. isotopes
c. electrons
d protons and neutrons
In the following molecule, label a portion that shows only primary structure; label two types of secondary structure; circle the tertiary structure.
The type of chemical bond between atoms with nearly equal electronegativities is called a(n) _____________ bond.
Shown is the amino acid tryptophan. Put the letter “C” at the site of every carbon atom. Label the amino group, the carboxyl group, and the side group.