Groups of atoms such as NH₂ or OH that appear in certain common arrangements are called __________ .
Ch. 2 - The Chemistry of Microbiology

Bauman6th EditionMicrobiology with Diseases by TaxonomyISBN: 9780134832302Not the one you use?Change textbook
Chapter 2, Problem 5
Which of the following terms most correctly describes the bonds in a molecule of water?
a. Nonpolar covalent bond
b. Polar covalent bond
c. Ionic bond
d. Hydrogen bond
 Verified step by step guidance
Verified step by step guidance1
Step 1: Understand the types of bonds listed in the options. A nonpolar covalent bond involves equal sharing of electrons between atoms, while a polar covalent bond involves unequal sharing of electrons, creating partial charges.
Step 2: Recall the structure of a water molecule (H₂O). It consists of one oxygen atom bonded to two hydrogen atoms. Oxygen is more electronegative than hydrogen, meaning it attracts electrons more strongly.
Step 3: Because oxygen attracts electrons more strongly, the electrons in the O-H bonds are shared unequally, resulting in a partial negative charge near oxygen and partial positive charges near the hydrogens.
Step 4: This unequal sharing of electrons in the O-H bonds classifies these bonds as polar covalent bonds, not nonpolar covalent, ionic, or hydrogen bonds (hydrogen bonds occur between molecules, not within a single molecule).
Step 5: Therefore, the correct term to describe the bonds within a water molecule is 'polar covalent bond'.

Verified video answer for a similar problem:
This video solution was recommended by our tutors as helpful for the problem above.
Video duration:
4mWas this helpful?
Key Concepts
Here are the essential concepts you must grasp in order to answer the question correctly.
Polar Covalent Bonds
Polar covalent bonds occur when two atoms share electrons unequally due to differences in electronegativity, resulting in partial positive and negative charges within the molecule. In water, oxygen is more electronegative than hydrogen, causing the shared electrons to be pulled closer to oxygen, creating polarity.
Recommended video:
Guided course
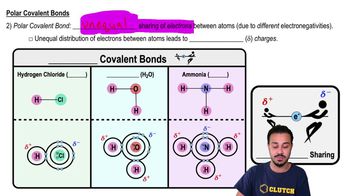
Polar Covalent Bonds
Nonpolar Covalent Bonds
Nonpolar covalent bonds involve equal sharing of electrons between atoms with similar electronegativities, resulting in no significant charge separation. Molecules like O2 or N2 typically have nonpolar covalent bonds, unlike water where the bond is polar.
Recommended video:
Guided course

Nonpolar Covalent Bonds
Hydrogen Bonds
Hydrogen bonds are weak intermolecular attractions between a hydrogen atom covalently bonded to an electronegative atom (like oxygen) and another electronegative atom nearby. In water, hydrogen bonds form between molecules, not within the bonds holding the atoms together.
Recommended video:
Guided course

Hydrogen Bonding
Related Practice
Textbook Question
918
views
Textbook Question
In water, cations and anions of salts dissociate from one another and become surrounded by water molecules. In this state, the ions are also called __________ .
a. electrically negative
b. ionically bonded
c. electrolytes
d. hydrogen bonds
1445
views
Textbook Question
Which of the following is not an organic compound?
a. Monosaccharide
b. Formaldehyde
c. Water
d. Steroid
1696
views
Textbook Question
Which of the following can be most accurately described as a decomposition reaction?
a. C₆H₁₂O₆ + 6 O₂ → 6 H₂O + 6 CO₂
b. glucose + ATP → glucose phosphate + ADP
c. 6 H₂O + 6 CO₂ → C₆H₁₂O₆ + 6 O₂
d. A + BC → AB + C
742
views
Textbook Question
The reverse of dehydration synthesis is __________ .
1352
views
Textbook Question
Explain how the polarity of water molecules makes water an excellent solvent.
1839
views
