 Back
BackControlling Microbial Growth in the Body: Antimicrobial Agents and Resistance
Study Guide - Smart Notes

Controlling Microbial Growth in the Body
Introduction to Antimicrobial Agents
Antimicrobial agents are essential tools in modern medicine for controlling microbial growth within the human body. Their effectiveness relies on selective toxicity, which allows them to target pathogens without harming the host.
Selective toxicity: The ability of a drug to kill or inhibit microbes without damaging host cells.
Chemotherapeutic agents: Drugs used to treat diseases, including antimicrobials for infections.
Incineration: Not suitable for internal use; drugs are required for internal microbial control.
History and Sources of Antimicrobial Agents
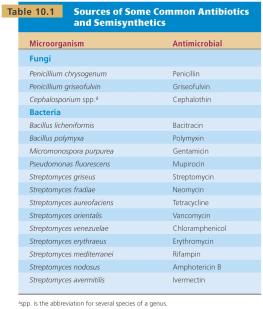
The development of antimicrobial agents revolutionized medicine, drastically reducing mortality from infectious diseases. Most antimicrobials are secondary metabolites produced by fungi and bacteria.
Salvarsan (arsphenamine): First modern chemotherapeutic agent (1910).
Penicillin: Discovered by Alexander Fleming (1929), produced by Penicillium mold.
Sulfanilamide: First widely available practical antimicrobial agent (1932).


Key Factors for Antimicrobial Action
The effectiveness of antimicrobial drugs depends on their ability to selectively target pathogens.
Antibacterial drugs: Largest group of antimicrobials.
Antifungal, antiprotozoan, antihelminthic, antiviral drugs: Fewer options due to similarities between eukaryotic pathogens and host cells.
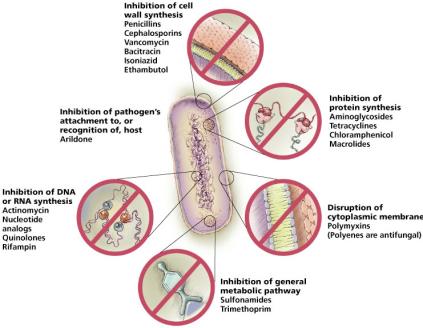
Mechanisms of Antimicrobial Action
Inhibition of Cell Wall Synthesis
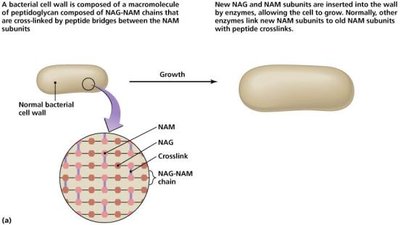
Drugs targeting cell wall synthesis exploit the unique structure of bacterial cell walls, particularly peptidoglycan.
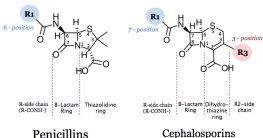
Beta-lactams: Prevent cross-linkage of NAM subunits, weakening cell walls and causing lysis.
Semisynthetic derivatives: More stable, better absorbed, broader spectrum.
Effectiveness: Only on growing cells; no effect on plant or animal cells.
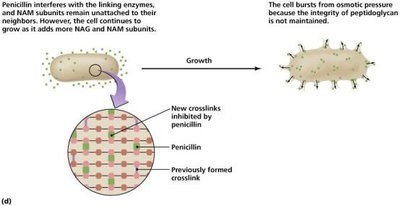
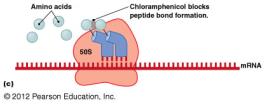
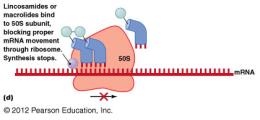
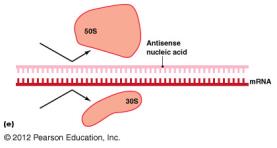
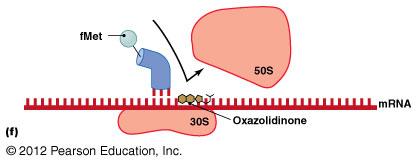
Inhibition of Protein Synthesis
Protein synthesis inhibitors target differences between prokaryotic (70S) and eukaryotic (80S) ribosomes.
Aminoglycosides: Cause misreading of mRNA (e.g., streptomycin).
Tetracyclines: Block tRNA docking site.
Chloramphenicol: Blocks peptide bond formation.
Macrolides: Block mRNA movement through ribosome.
Antisense RNA: Prevents ribosome assembly.
Oxazolidinones: Block initiation of translation.
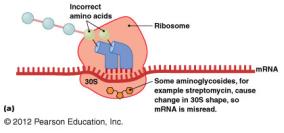
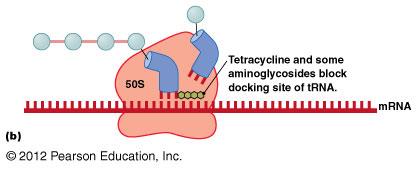
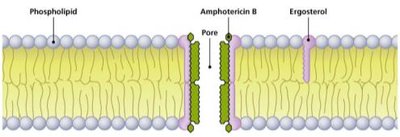
Disruption of Cytoplasmic Membranes
Some drugs disrupt membrane integrity, particularly in fungi.
Amphotericin B: Binds to ergosterol in fungal membranes, forming pores.
Selective toxicity: Humans are somewhat susceptible due to cholesterol similarity.
Inhibition of Metabolic Pathways
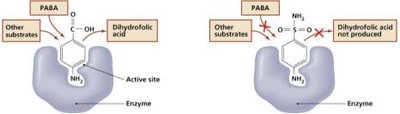
Antimetabolic agents target differences in metabolic processes between pathogens and hosts.
Heavy metals: Inactivate enzymes.
Metabolic antagonists: Inhibit biosynthesis of DNA and RNA nucleotides (e.g., sulfa drugs).
Enzyme inhibitors: Block activation of viruses or paralyze parasitic worms.
Nucleic Acid Analogs
Nucleic acid analogs mimic nucleotides, causing premature termination of nucleic acid synthesis.
Acyclovir: Used for herpes, terminates DNA replication.
Remdesivir: Used for COVID-19, terminates RNA replication.
Retrovir: Used for HIV, terminates RNA replication.
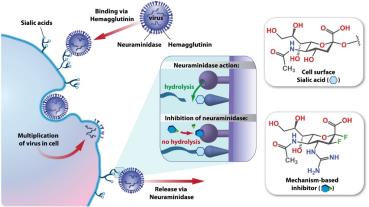
Inhibition of Viral Attachment and Release
Drugs can prevent viruses from attaching to or releasing from host cells.
Neuraminidase inhibitors (Relenza, Tamiflu): Block release of influenza virions by mimicking sialic acid.
Spectrum of Action and Efficacy
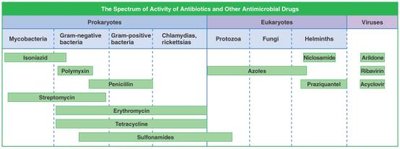
Spectrum of Activity
Antimicrobials vary in their spectrum of activity, affecting different groups of organisms.
Broad-spectrum drugs: Affect a wide range of organisms but may cause superinfections.
Narrow-spectrum drugs: Target specific groups.
Efficacy Testing
The effectiveness of antibacterial agents is determined by laboratory tests.
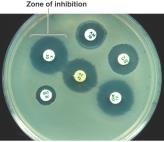
Disk-diffusion (Kirby-Bauer) test: Measures zone of inhibition.
Minimum inhibitory concentration (MIC): Lowest concentration preventing growth.
Minimum bactericidal concentration (MBC): Lowest concentration killing bacteria.


Administration, Safety, and Side Effects
Routes of Administration
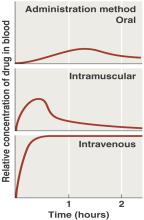
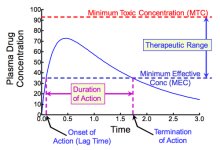
The route of administration affects drug concentration and efficacy.
Topical: Direct application to skin.
Oral: Simple, but compliance issues.
Intramuscular (IM): Injection into muscle.
Intravenous (IV): Direct injection into bloodstream; fastest and highest concentration.
Safety and Side Effects
Antimicrobial drugs can cause toxicity, allergies, and disruption of normal flora.
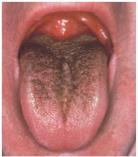
Toxicity: Can affect kidneys, liver, nerves; tetracycline damages teeth and bones.
Allergies: Rare but potentially life-threatening.
Disruption of normal flora: Can lead to yeast infections or pseudomembranous colitis.
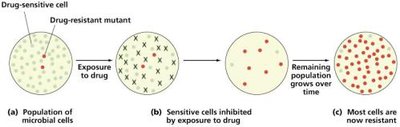
Development and Mechanisms of Resistance
Acquisition of Resistance
Bacteria acquire resistance through mutations or horizontal gene transfer.
Spontaneous mutations: Changes in chromosomal genes.
Antibiotic resistance plasmids: Acquired via transformation, transduction, conjugation.
Population resistance: Occurs when resistant microbes become the majority.
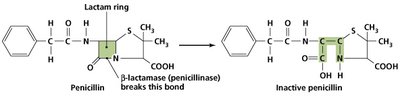
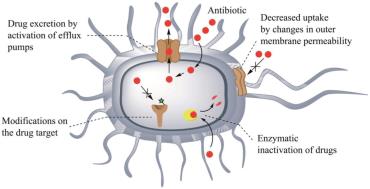
Bacterial Resistance Mechanisms
Bacteria employ several strategies to resist antimicrobial drugs.
Enzyme production: Inactivates or destroys antibiotics (e.g., beta-lactamase).
Prevention of drug entry: Changes in membrane proteins.
Alteration of drug receptor: Modifies target site.
Alteration of metabolic chemistry: Changes metabolic pathways.
Multidrug resistance proteins: Efflux pumps expel drugs.
Methods for Retarding Resistance
Strategies to slow resistance development include proper drug use and combination therapy.
Maintain high drug concentrations: Ensures all sensitive cells are killed.
Combination therapy: Synergistic effects reduce resistance.
Limit use: Only use antimicrobials when necessary.
Develop new drugs: Modify existing drugs for greater efficacy.
Summary Table: Mechanisms of Antimicrobial Action
Mechanism | Example Drugs | Target |
|---|---|---|
Inhibition of Cell Wall Synthesis | Penicillins, Cephalosporins, Vancomycin | Peptidoglycan |
Inhibition of Protein Synthesis | Aminoglycosides, Tetracyclines, Macrolides | Ribosomes |
Disruption of Cytoplasmic Membrane | Polymyxins, Amphotericin B | Membrane integrity |
Inhibition of Metabolic Pathways | Sulfonamides, Trimethoprim | Enzymes |
Inhibition of Nucleic Acid Synthesis | Actinomycin, Nucleotide analogs | DNA/RNA polymerases |
Inhibition of Attachment/Release | Arildone, Neuraminidase inhibitors | Viral proteins |
Conclusion
Understanding the mechanisms, efficacy, and resistance of antimicrobial agents is crucial for their effective use in controlling microbial growth and preventing the spread of resistant organisms. Additional info: Academic context was added to clarify mechanisms, historical significance, and clinical implications of antimicrobial agents and resistance.
