 Back
BackCh 9 - Controlling Microbial Growth in the Environment
Study Guide - Smart Notes

Controlling Microbial Growth in the Environment
Introduction
Microbial control in the environment is essential for preventing infection, ensuring food safety, and maintaining sterile conditions in medical and laboratory settings. This chapter covers the principles, methods, and applications of physical and chemical agents used to control microbial growth.
Basic Principles of Microbial Control
Terminology of Microbial Control
Understanding the terminology is crucial for distinguishing between different microbial control strategies. The following table summarizes key terms, their definitions, examples, and comments:
Term | Definition | Examples | Comments |
|---|---|---|---|
Antisepsis | Reduction in the number of microorganisms and viruses, particularly potential pathogens, on living tissue | Use of iodine or alcohol to prepare skin for injection | Antiseptics are frequently disinfectants whose strength has been reduced for use on living tissue |
Aseptic | Refers to an environment or procedure free of pathogenic contaminants | Preparation of surgical field; hand washing; flame sterilization of laboratory equipment | Scientists, laboratory technicians, and health care workers routinely follow aseptic techniques |
-cide/-cidal | Suffixes indicating destruction of a type of microbe | Bactericide; fungicide; germicide; virucide | Germicides include ethylene oxide, propylene oxide, and aldehydes |
Degerming | Removal of microbes by mechanical means | Handwashing; alcohol swabbing at site of injection | Chemicals play a secondary role to the mechanical removal of microbes |
Disinfection | Destruction of most microorganisms and viruses on nonliving tissue | Use of phenolics, alcohols, aldehydes, or soaps on equipment or surfaces | Term is used primarily in relation to pathogens |
Pasteurization | Use of heat to destroy pathogens and reduce the number of spoilage microorganisms in foods and beverages | Pasteurized milk and fruit juices | Heat treatment is brief to avoid altering taste and nutrients; microbes still remain and eventually cause spoilage |
Sanitization | Removal of pathogens from objects to meet public health standards | Washing tableware in scalding water in restaurants | Standards of sanitization vary among governmental jurisdictions |
-stasis/-static | Suffixes indicating inhibition, but not complete destruction, of a type of microbe | Bacteriostatic; fungistatic; virustatic | Germistats include some chemicals, refrigeration, and freezing |
Sterilization | Destruction of all microorganisms and viruses in or on an object | Preparation of microbiological culture media and canned food | Typically achieved by steam under pressure, incineration, or by ethylene oxide gas |

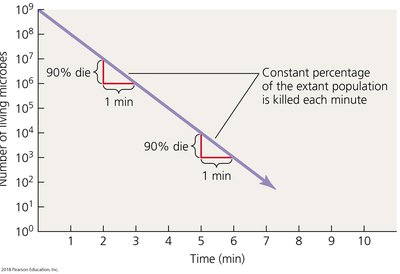
Microbial Death Rates
Microbial death is defined as the permanent loss of reproductive ability under ideal environmental conditions. The microbial death rate is a measurement of the efficacy of an antimicrobial agent, indicating the constant percentage of the extant population killed per unit time.
Significance: Understanding death rates helps in designing effective sterilization and disinfection protocols.
Mathematical Representation: Microbial death often follows a logarithmic pattern, where a constant proportion of cells die per unit time.
Action of Antimicrobial Agents
Antimicrobial agents act by targeting essential cellular structures and functions:
Cell Walls: Damage leads to osmotic lysis.
Cytoplasmic Membranes: Disruption causes leakage of cellular contents.
Proteins: Denaturation by heat or chemicals disrupts function.
Nucleic Acids: Damage by chemicals, radiation, or heat can produce fatal mutations or halt protein synthesis.

The Selection of Microbial Control Methods
Factors Affecting the Efficacy of Antimicrobial Methods
Several factors influence the choice and effectiveness of microbial control methods:
Site to be Treated: The nature of the surface or tissue determines the method (e.g., harsh chemicals cannot be used on living tissue).
Relative Susceptibility of Microorganisms: Microbes vary in resistance; prions, endospores, and protozoan cysts are among the most resistant.
Environmental Conditions: Temperature, pH, and presence of organic materials can enhance or inhibit antimicrobial efficacy.
Levels of Germicides
High-level germicides: Kill all pathogens, including endospores.
Intermediate-level germicides: Kill fungal spores, protozoan cysts, viruses, and pathogenic bacteria.
Low-level germicides: Kill vegetative bacteria, fungi, protozoa, and some viruses.
Biosafety Levels
Laboratories are classified into four biosafety levels (BSL) based on the risk associated with the pathogens handled:
BSL-1: Non-pathogenic microbes.
BSL-2: Moderately hazardous agents.
BSL-3: Pathogens handled in safety cabinets.
BSL-4: Dangerous and exotic microbes causing severe or fatal diseases.

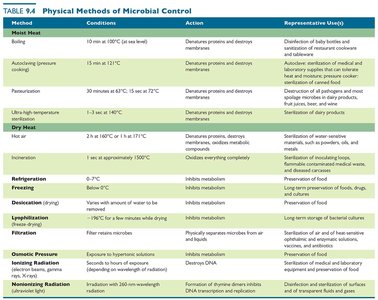
Physical Methods of Microbial Control
Overview of Physical Methods
Physical methods include heat, cold, desiccation, filtration, osmotic pressure, and radiation. Each method has specific applications and limitations.
Heat-Related Methods
Moist Heat: Denatures proteins and destroys membranes. Methods include boiling, autoclaving, pasteurization, and ultra-high-temperature sterilization.
Dry Heat: Used for materials that cannot be sterilized with moist heat; requires higher temperatures and longer times.
Key Definitions
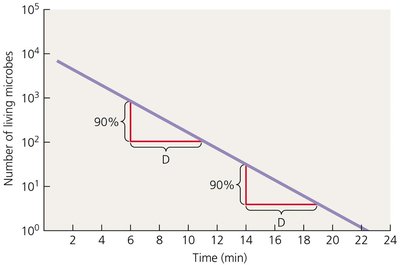
Thermal Death Point: Lowest temperature that kills all cells in 10 minutes.
Thermal Death Time: Time to sterilize a volume of liquid at a set temperature.
Decimal Reduction Time (D): Time required to destroy 90% of microbes in a sample.
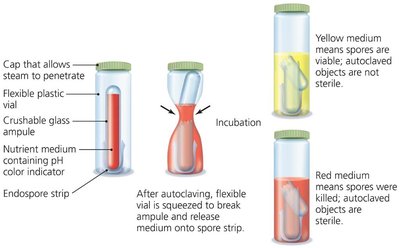
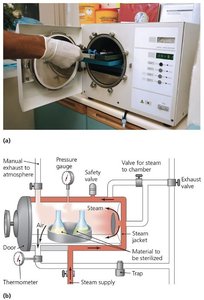
Autoclaving and Sterility Indicators
Autoclaving uses pressurized steam (121°C, 15 psi, 15 min) to achieve sterilization. Sterility indicators, such as endospore strips, confirm successful sterilization.
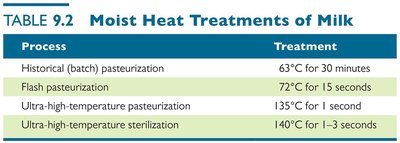
Pasteurization Methods
Pasteurization reduces microbial load in foods and beverages without sterilizing. Methods include batch, flash, and ultra-high-temperature pasteurization.
Process | Treatment |
|---|---|
Historical (batch) pasteurization | 63°C for 30 minutes |
Flash pasteurization | 72°C for 15 seconds |
Ultra-high-temperature pasteurization | 135°C for 1 second |
Ultra-high-temperature sterilization | 140°C for 1–3 seconds |
Refrigeration and Freezing
Slows microbial metabolism and growth.
Some microbes can survive or multiply at low temperatures.
Slow freezing is more effective than quick freezing.
Desiccation and Lyophilization
Desiccation: Inhibits growth by removing water.
Lyophilization: Freeze-drying for long-term preservation; prevents ice crystal formation.

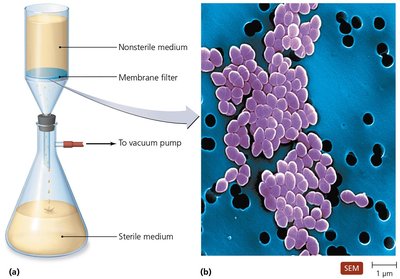
Filtration
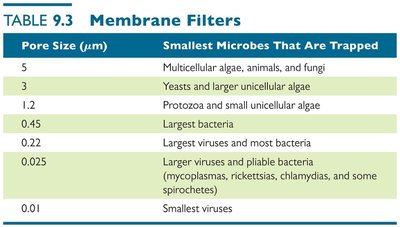
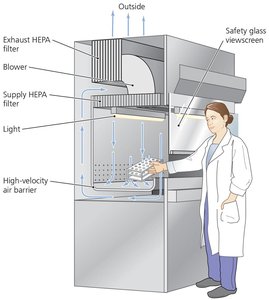
Filtration physically removes microbes from air or liquids using membrane filters of defined pore sizes. HEPA filters are used in safety cabinets and hospital air systems.
Pore Size (µm) | Smallest Microbes That Are Trapped |
|---|---|
5 | Multicellular algae, animals, and fungi |
3 | Yeasts and larger unicellular algae |
1.2 | Protozoa and small unicellular algae |
0.45 | Largest bacteria |
0.22 | Largest viruses and most bacteria |
0.025 | Larger viruses and pliable bacteria |
0.01 | Smallest viruses |
Osmotic Pressure
High concentrations of salt or sugar create hypertonic environments, causing cells to lose water and inhibiting growth.
Fungi are more tolerant of hypertonic conditions than bacteria.
Radiation
Ionizing Radiation: (e.g., gamma rays, X-rays) Ejects electrons, creating ions that damage DNA and proteins. Used for sterilizing medical equipment and food.
Nonionizing Radiation: (e.g., UV light) Causes DNA damage (pyrimidine dimers); used for disinfecting air, surfaces, and transparent fluids.

Chemical Methods of Microbial Control
Overview of Chemical Methods
Chemical agents target cell walls, membranes, proteins, or DNA. Their effectiveness depends on environmental conditions and the type of microbe.
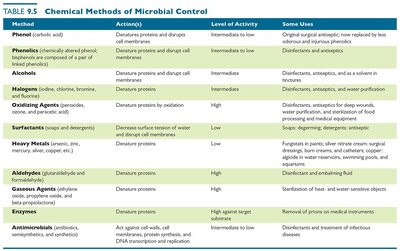
Method | Action(s) | Level of Activity | Some Uses |
|---|---|---|---|
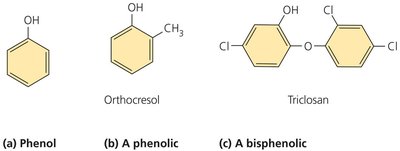
Phenol | Denatures proteins and disrupts cell membranes | Intermediate to low | Original surgical antiseptic, now replaced by less odorous and injurious phenolics |
Phenolics | Denature proteins and disrupt cell membranes | Intermediate to low | Disinfectants and antiseptics |
Alcohols | Denature proteins and disrupt cell membranes | Intermediate | Disinfectants, antiseptics, and as a solvent in tinctures |
Halogens | Denature proteins | Intermediate | Disinfectants, antiseptics, and water purification |
Oxidizing Agents | Denature proteins by oxidation | High | Disinfectants, antiseptics for deep wounds, water purification, and sterilization of food-processing and medical equipment |
Surfactants | Decrease surface tension of water and disrupt cell membranes | Low | Soaps, detergents, and antiseptics |
Heavy Metals | Denature proteins | Low | Fungistats in paints, silver nitrate cream, surgical dressings, burn creams, and catheters |
Aldehydes | Denature proteins | High | Disinfectant and embalming fluid |
Gaseous Agents | Denature proteins and DNA by cross-linking functional groups | High | Sterilization of heat- and water-sensitive objects |
Enzymes | Denature proteins | High against target substrate | Removal of prions on medical instruments |
Antimicrobials | Act against cell walls, cell membranes, protein synthesis, and DNA transcription and replication | Intermediate to low | Disinfectants and treatment of infectious diseases |
Phenol and Phenolics
Denature proteins and disrupt cell membranes.
Effective in the presence of organic matter and remain active for prolonged periods.
Commonly used in healthcare settings, but have a strong odor and potential side effects.
Alcohols
Intermediate-level disinfectants that denature proteins and disrupt membranes.
Most effective at concentrations of 70–90%.
Used for skin antisepsis and instrument disinfection.
Halogens
Intermediate-level agents that damage enzymes by denaturation.
Include iodine, chlorine, bromine, and fluorine compounds.
Used for water disinfection, antiseptics, and surface cleaning.
Oxidizing Agents
High-level disinfectants and antiseptics (e.g., hydrogen peroxide, ozone, peracetic acid).
Kill by oxidation of microbial enzymes.
Used for sterilizing equipment and treating water.
Surfactants
Reduce surface tension of solvents, aiding in mechanical removal of microbes.
Soaps are good degerming agents; detergents (quats) disrupt membranes and are low-level disinfectants.
Heavy Metals
Denature proteins; low-level bacteriostatic and fungistatic agents.
Examples: silver nitrate, thimerosal, copper sulfate.
Aldehydes
Compounds with terminal –CHO groups; cross-link functional groups to denature proteins and inactivate nucleic acids.
Glutaraldehyde and formaldehyde are common examples.
Gaseous Agents
Used in closed chambers to sterilize items; denature proteins and DNA.
Hazardous, potentially explosive, and carcinogenic.
Enzymes
Antimicrobial enzymes (e.g., lysozyme, prionzyme) target specific microbial structures.
Used in food processing and medical instrument sterilization.
Antimicrobial Drugs
Include antibiotics, semisynthetics, and synthetics.
Primarily used for disease treatment, but some are used for environmental control.
Methods for Evaluating Disinfectants and Antiseptics
Evaluation Methods
Phenol Coefficient: Compares efficacy to phenol; values >1 indicate greater effectiveness.
Use-Dilution Test: Measures effectiveness against specific microbes on metal cylinders.
Kelsey-Sykes Capacity Test: Assesses minimum time required for a disinfectant to be effective.
In-Use Test: Monitors effectiveness in real-world conditions by sampling surfaces before and after disinfection.
Development of Resistant Microbes
Overuse of antiseptics and disinfectants can promote the development of resistant microbial strains, reducing the effectiveness of these agents over time.
