 Back
BackDefining a Healthy Gut Microbiome: Concepts, Variability, and Clinical Implications
Study Guide - Smart Notes

The Healthy Gut Microbiome
Introduction to the Microbiome
The microbiome refers to the collective genetic material of all microorganisms (bacteria, viruses, fungi, archaea) residing in and on the human body. Understanding what constitutes a 'healthy' gut microbiome is crucial for developing microbiome-based diagnostics and therapies. However, defining a healthy microbiome is complex due to vast interindividual variability and the influence of numerous factors such as genetics, environment, diet, and lifestyle.
Determinants of Microbiome Composition
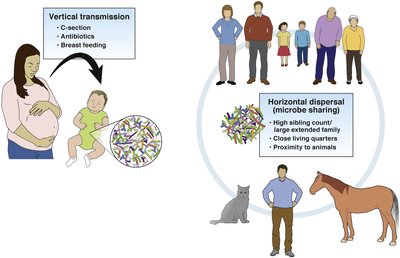
Transmission and Modifiers
Vertical transmission: Microbes are passed from mother to infant during birth and breastfeeding.
Horizontal transmission: Microbes are exchanged between individuals and the environment, influenced by cohabitation, pets, and living conditions.
Personal factors: Genetics, birth mode (vaginal vs. C-section), age, gender, and migration history affect microbiome composition.
Environmental and lifestyle factors: Socio-economic status, diet, and medication use are major modifiers.
Defining Health and Normality in the Microbiome
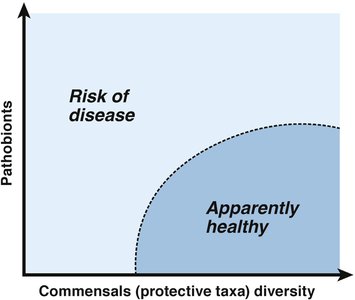
Conceptual Challenges
There is no universally accepted definition of a 'healthy' microbiome. Health is a relative condition, and 'normal' does not always equate to 'healthy.' Studies show that there is no single microbiome configuration required for health; instead, multiple configurations can be consistent with health. The emergence of disease is context-dependent and influenced by the balance between protective commensals and potentially harmful pathobionts.
Microbiome Variability and Population Differences
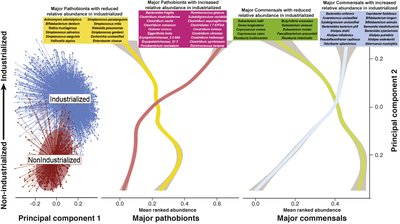
Industrialized vs. Nonindustrialized Microbiomes
Microbiome composition varies significantly between populations in industrialized and nonindustrialized societies. These differences are shaped by diet, lifestyle, and environmental exposures. Industrialized microbiomes are often adapted for mucin degradation and reflect exposure to antibiotics and medications, while nonindustrialized microbiomes are adapted for fiber degradation.
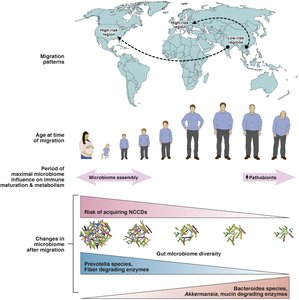
Migration and Microbiome Shifts
Migration from nonindustrialized to industrialized regions is associated with increased risk of noncommunicable chronic diseases (NCCDs) such as Crohn’s disease, asthma, and type 2 diabetes. The risk is highest in those migrating during early childhood, when the microbiome is being assembled and immune system maturation is most influenced by microbial signals. Migration leads to a progressive shift in microbiome composition toward that of the host country.
Classifying Microbiomes
Core Microbiome and Functional Redundancy
Core microbiome: Originally thought to be a set of microbes required for health, but now understood as a set of core functions, which vary by context.
Functional redundancy: Different bacteria can perform similar metabolic functions, making metabolic activity more conserved than taxonomic composition.
This suggests that functional (metabolomic and metaproteomic) definitions may be superior to purely taxonomic ones for distinguishing healthy from abnormal microbiomes.
Lessons from Outliers
Ethnic Minorities and Microbiome Diversity
Outlier populations, such as the Irish Travellers, retain a nonindustrialized-type microbiome despite living in an industrialized country. This is attributed to lifestyle factors such as large family sizes, close living quarters, and proximity to animals, which promote horizontal microbe sharing. These outliers challenge restrictive definitions of a healthy microbiome and highlight the importance of nondietary determinants.

Strain-Level Variation
Importance of Strain Resolution
Strain-level differences within species can have significant functional and clinical implications.
For example, Escherichia coli strains range from beneficial to pathogenic.
Advanced sequencing technologies (shotgun metagenomics) now allow for strain-level discrimination, which is critical for understanding functional capacity and disease associations.
Microbiome Dynamics Across the Lifespan
Age-Related Changes and Disease Risk
The gut microbiome assembles during the first three years of life, influencing immune and metabolic maturation. In later life, aging, disease, and medications can destabilize the microbiome, leading to increased risk of chronic diseases. Loss of dietary diversity in the elderly is associated with reduced microbiota diversity and increased inflammation and frailty. Age-related changes in the microbiome can overlap with disease-associated changes, complicating the identification of disease-specific biomarkers.
Beyond Bacteria: The Dark Matter of the Microbiome
Mycobiome, Virome, and Archaeome
Many components of the microbiome, such as viruses (virome), fungi (mycobiome), and archaea (archaeome), are poorly characterized.
Bacteriophages (viruses that infect bacteria) may play important roles in modulating the bacterial microbiome and influencing health outcomes.
Comprehensive studies integrating bacteria, phage, fungi, and metabolites are needed to fully understand the microbiome’s impact on health and disease.
Clinical Implications and Future Directions
Microbiome-Based Diagnostics and Therapies
Microbiome profiles can serve as biomarkers for disease risk, prognosis, and therapy response, but interpretation must consider individual variability and context.
Fecal microbial transplantation (FMT) is effective for recurrent Clostridioides difficile infection and is being explored for other indications.
Dietary interventions can modify the microbiome and may help prevent or delay age-related diseases.
Antimicrobial resistance is a major challenge; the microbiome may provide alternatives to antibiotics, such as bacteriophages and antimicrobial metabolites.
Limitations and Communication
Technical limitations in microbiome profiling and the need for larger, more diverse cohorts remain significant challenges.
Clear communication of microbiome science to the public is essential to avoid misunderstandings and overinterpretation of results.
Summary Table: Factors Influencing the Gut Microbiome
Factor | Examples | Impact |
|---|---|---|
Genetics | Host lineage, ethnicity | Shapes susceptibility to colonization by specific taxa |
Birth Mode | Vaginal vs. C-section | Initial microbial exposure and colonization patterns |
Diet | Fiber-rich vs. Western diet | Promotes different microbial communities and functions |
Medications | Antibiotics, metformin, statins | Alter microbiome composition and function |
Environment | Urban vs. rural, cohabitation, pets | Facilitates horizontal transmission and diversity |
Age | Infancy, adulthood, elderly | Dynamic changes in composition and function |
Lifestyle | Migration, socioeconomic status | Associated with shifts in microbiome and disease risk |
Additional info: The notes above integrate and expand upon the provided material with academic context, including definitions, examples, and clinical implications relevant to college-level microbiology students.
