 Back
BackDNA & RNA Viral Pathogens: Diseases by Body System
Study Guide - Smart Notes

Major Human Viral Diseases: Overview
Classification by Body System
Viral pathogens are classified based on the primary body system they affect. This classification aids in understanding disease mechanisms, transmission, and clinical management.
Respiratory Viruses: Influenza virus, SARS-CoV-2, Measles virus
Blood/Immune System Viruses: HIV
Skin & Nerve System Viruses: HSV-1, HSV-2, Human papillomavirus (HPV)
Gastrointestinal Viruses: Norovirus
Principles of Antiviral Therapy
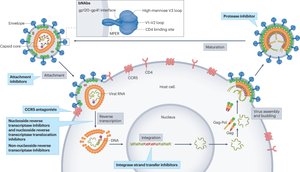
Mechanisms of Action
Antiviral drugs are designed to target specific steps in the viral life cycle. Because viruses rely on host cell machinery, achieving selective toxicity is more challenging than with antibacterial agents.
Attachment/Entry Inhibitors: Block viral binding or fusion with host cells.
Uncoating Inhibitors: Prevent release of viral genome into the host cell.
Polymerase/Reverse Transcriptase Inhibitors: Inhibit viral genome replication.
Integrase Inhibitors: Block integration of viral DNA into host genome (e.g., HIV).
Protease Inhibitors: Prevent maturation of viral proteins.
Release/Budding Inhibitors: Block release of new virions from host cells.
Most antivirals suppress viral replication but do not eliminate latent infections.
Viral Diseases of the Respiratory System
General Features
The respiratory tract is a major portal for viral entry, replication, and transmission. Viruses may infect the upper or lower respiratory tract and are primarily spread by droplets and aerosols.
Replication occurs in respiratory epithelial cells.
Disease results from both viral replication and host inflammatory response.
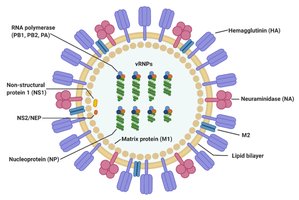
Influenza Virus
Influenza viruses are enveloped, negative-sense single-stranded RNA viruses (family Orthomyxoviridae) with a segmented genome. Their surface proteins, hemagglutinin (HA) and neuraminidase (NA), are critical for viral entry and release, respectively.
Genome: 8 RNA segments allow for rapid evolution via reassortment.
HA: Mediates attachment and entry by binding sialic acid receptors.
NA: Facilitates release of new virions from infected cells.
Pathogenesis
HA binds sialic acid receptors on epithelial cells, enabling viral entry.
Replication leads to cell death and loss of mucociliary clearance.
Infected cells release cytokines (e.g., IL-1, TNF-α, IFNs), causing inflammation.
Symptoms: Fever, cough, and increased susceptibility to secondary infections.
Antigenic Drift vs. Antigenic Shift
Antigenic Drift: Gradual accumulation of point mutations in HA/NA, leading to seasonal variation.
Antigenic Shift: Abrupt reassortment of genome segments, creating new viral strains with pandemic potential.
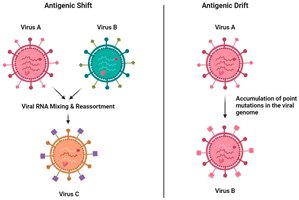
Immunity and Vaccination
Annual vaccination is necessary due to antigenic drift, which alters viral antigens and reduces the effectiveness of previous immune responses.
SARS-CoV-2 (COVID-19)
SARS-CoV-2 is an enveloped, positive-sense single-stranded RNA virus (family Coronaviridae). The spike (S) protein binds to the ACE2 receptor on pulmonary epithelial cells, mediating viral entry and serving as a major immune target.
Causes COVID-19, a respiratory disease with variable severity.
Transmission: Droplets and aerosols.
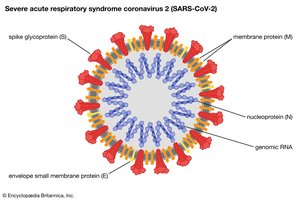
Pathogenesis
Spike protein binds ACE2, allowing viral entry and replication in respiratory epithelial cells.
Cytokine release (e.g., IL-6, IFNs) leads to inflammation.
Severe cases involve immune dysregulation and lung damage.
Influenza vs. COVID-19: Epidemiology
Feature | Influenza | COVID-19 |
|---|---|---|
Incubation Time | 1–4 days | 2–14 days |
Transmission | Droplets/aerosols | Droplets/aerosols |
Asymptomatic Spread | Rare | Common |
Hospitalization Rate | ~2% | ~19% |
Case Fatality Rate | 0.1% or less | 0.3–4% |
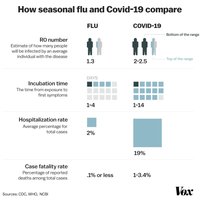
Viral Diseases of the Immune System
HIV (Human Immunodeficiency Virus)
HIV is an enveloped, positive-sense single-stranded RNA retrovirus (family Retroviridae) that infects CD4+ T cells and macrophages. It uses reverse transcriptase to integrate its genome into host DNA, causing progressive immune system damage.
gp120: Binds to host cell receptor (CD4).
gp41: Mediates viral membrane fusion.
Reverse Transcriptase: Converts viral RNA into DNA for integration.
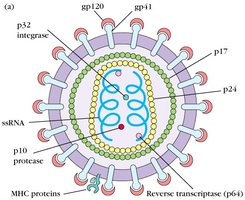
HIV Lifecycle
Attachment and entry
Reverse transcription
Nuclear import
Integration into host genome
Transcription and translation
Assembly, budding, and maturation
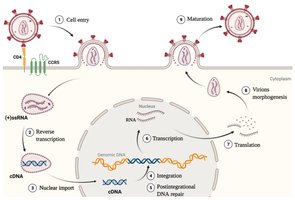
Pathogenesis
Progressive loss of CD4+ T cells leads to immunodeficiency (AIDS).
Latent reservoirs in long-lived T cells make eradication difficult.
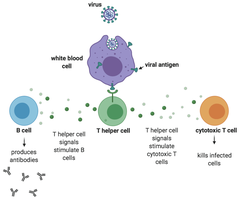
Transmission & Prevention
Spread via blood, sexual fluids, and vertical transmission (mother to child).
Prevention: Safe sex, needle control, screening, and antiretroviral drugs (ART, PrEP).
ART suppresses viral replication, preserves CD4+ T cells, and slows progression to AIDS but does not eliminate latent reservoirs.
Viral Diseases of the Skin & Nervous System
Latency and Reactivation
Some viruses establish lifelong latent infections in neurons, with viral DNA persisting in host cells and reactivating under certain conditions.
Latency: Viral DNA persists with minimal gene expression and no virion production.
Reactivation: Triggered by stress, immune suppression, or environmental cues.
Herpes Simplex Viruses (HSV-1 & HSV-2)
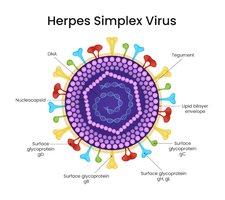
HSV-1 and HSV-2 are enveloped, double-stranded DNA viruses (family Herpesviridae) that infect epithelial cells and sensory neurons, establishing lifelong latency.
HSV-1: Primarily causes oral lesions.
HSV-2: Primarily causes genital lesions.
Tegument: Protein-rich layer between envelope and capsid, important for early infection events.
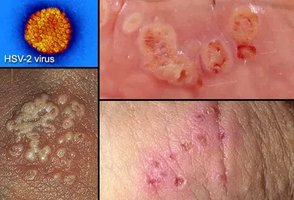
HSV Treatment
Spread by direct contact with lesions.
Prevention: Barrier protection, avoiding contact during outbreaks.
Drug: Acyclovir inhibits viral DNA polymerase, reducing severity and recurrence but not curing latency.
Varicella-Zoster Virus (VZV)
VZV causes chickenpox in children and shingles in adults. The virus establishes latency in sensory neurons and can reactivate later in life.
Chickenpox: Primary infection, usually in children.
Shingles: Reactivation in adults, often with painful rash.
Measles Virus (Rubeola)
Overview and Pathogenesis
Measles virus is an enveloped, negative-sense single-stranded RNA virus (family Paramyxoviridae) that spreads via respiratory droplets and infects immune cells, causing systemic infection.
Highly infectious (R0 = 12–18).
Causes characteristic rash and Koplik’s spots.
Destroys immune memory cells, leading to “immune amnesia.”
Prevention
MMR vaccine (live attenuated) induces long-term immunity.
High vaccination coverage is required for herd immunity.
Human Papillomavirus (HPV)
Overview and Pathogenesis
HPV is a non-enveloped, double-stranded DNA virus that infects basal epithelial cells. Low-risk types cause warts, while high-risk types (via E6 and E7 proteins) can lead to oncogenesis by disrupting tumor suppressor pathways.
E6 promotes degradation of p53 (blocks apoptosis).
E7 inactivates Rb (promotes cell cycle progression).
HPV and Transformation
HPV DNA integration disrupts E2 gene, increasing E6/E7 expression.
Loss of tumor suppressor control leads to uncontrolled cell growth and cancer risk.
Viral Diseases of the Gastrointestinal Tract
Norovirus
Norovirus is a non-enveloped, positive-sense single-stranded RNA virus that infects intestinal epithelial cells. It is highly contagious, environmentally stable, and causes acute vomiting and diarrhea.
Disrupts ion transport and blunts villi, leading to decreased absorption and “leaky gut.”
Spread via fecal–oral transmission, often causing outbreaks.
Summary Table: Major Viral Disease Mechanisms
Virus | Key Mechanism |
|---|---|
Respiratory viruses | Rapid transmission, high mutation rate |
HIV | Chronic infection, immune depletion |
HSV | Latency and reactivation |
Measles | Immune suppression (amnesia) |
HPV | Oncogenesis (E6/E7) |
GI viruses | High transmission, dehydration |
