 Back
BackInnate Immunity: Nonspecific Defenses of the Host – Inflammation, Fever, and Antimicrobial Substances
Study Guide - Smart Notes

Innate Immunity: Nonspecific Defenses of the Host
Overview of Innate Immunity
Innate immunity provides the first and second lines of defense against invading pathogens. It includes physical barriers, cellular defenses, inflammation, fever, and antimicrobial substances. These mechanisms act rapidly and are not specific to particular pathogens.
Component | Functions |
|---|---|
Phagocytes | Engulf and destroy microbes and debris (e.g., neutrophils, macrophages). |
Natural Killer (NK) cells | Kill infected or abnormal cells by releasing cytolytic granules. |
Inflammation | Localizes infection, recruits immune cells, and initiates tissue repair. |
Fever | Inhibits microbial growth and enhances immune responses. |
Complement system | Promotes cytolysis, opsonization, and inflammation. |
Interferons | Inhibit viral replication and activate immune cells. |
Iron-binding proteins | Limit microbial access to iron. |
Antimicrobial peptides (AMPs) | Directly kill microbes and modulate immune responses. |

Inflammation
Steps of Inflammation
Inflammation is a localized, protective response to tissue injury or infection. It aims to eliminate the initial cause of injury, clear out damaged cells, and establish tissue repair.
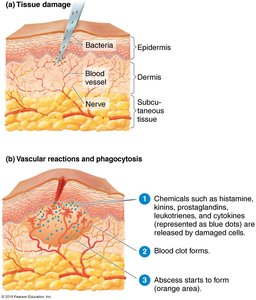
Step 1: Vasodilation and Increased Permeability – Blood vessels dilate and become more permeable, allowing defensive substances to enter the affected tissue. This is mediated by chemicals such as histamine, kinins, prostaglandins, leukotrienes, and cytokines.
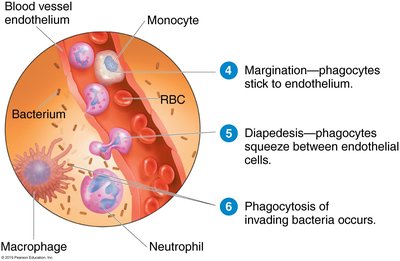
Step 2: Phagocyte Migration and Phagocytosis – Neutrophils and later macrophages migrate to the site, adhere to the endothelium (margination), squeeze through vessel walls (diapedesis), and ingest microbes and debris.
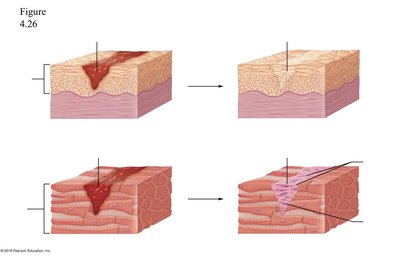
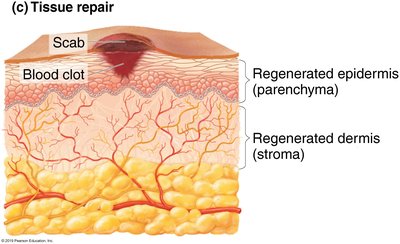
Step 3: Tissue Repair – Damaged tissue is repaired after harmful substances are removed. The stroma (supporting tissue) and parenchyma (functional tissue) may both be involved in repair.
Chemotaxis and Area Containment
Chemotaxis is the movement of phagocytes toward chemical signals released by damaged cells, microbes, and other immune cells. Clotting elements wall off the infected area to prevent the spread of pathogens.
Inflammation and Tissue Repair
Stroma: The supporting connective tissue; if active in repair, leads to fibrosis (scar formation).
Parenchyma: The functional tissue; if active, leads to regeneration (restoration of normal function).
Fever
Fever is an abnormally high body temperature, often in response to infection. The hypothalamus regulates body temperature and is reset by cytokines and prostaglandins during infection. Fever enhances immune responses and inhibits microbial growth.
Onset: Cytokines trigger the hypothalamus to raise the set point, causing vasoconstriction and shivering.
Crisis: When the infection subsides, the set point returns to normal, leading to vasodilation and sweating.
The Complement System
Overview
The complement system consists of over 30 serum proteins produced by the liver. These proteins enhance the ability of antibodies and phagocytic cells to clear microbes and damaged cells, promote inflammation, and attack the pathogen's plasma membrane.
Proteins are designated C1, C2, C3, etc., and are inactive until split into fragments (e.g., C3a, C3b).
Complement can be activated by three pathways: classical, alternative, and lectin.
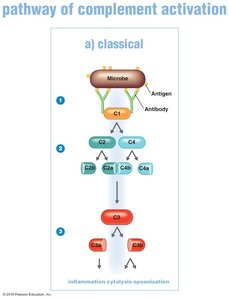
Classical Pathway
Initiated by antigen-antibody complexes, this pathway involves the sequential activation of complement proteins, leading to the cleavage of C3 and downstream effects.
Antibodies bind antigens, activating C1.
C1 splits and activates C2 and C4.
C2a and C4b combine to activate C3.
C3a promotes inflammation; C3b promotes cytolysis and opsonization.
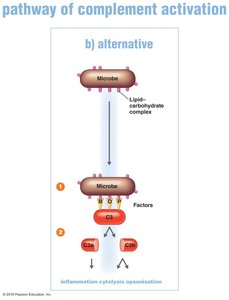
Alternative Pathway
This pathway is triggered directly by microbial surfaces without the need for antibodies.
C3 interacts with factors B, D, and P on the microbe surface.
C3 splits into C3a and C3b, with similar functions as in the classical pathway.
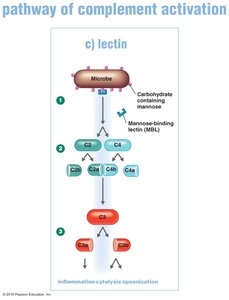
Lectin Pathway
Activated by mannose-binding lectin (MBL) binding to microbial carbohydrates, leading to complement activation similar to the classical pathway.
Macrophages release cytokines that stimulate lectin production in the liver.
MBL binds to mannose on the pathogen, activating C2 and C4, which then activate C3.
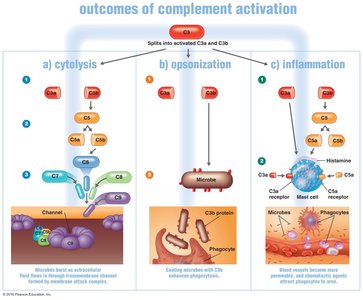
Outcomes of Complement Activation
Cytolysis: Formation of the membrane attack complex (MAC) that creates pores in microbial membranes, leading to cell lysis. Gram-negative bacteria are more susceptible.
Opsonization: C3b coats microbial surfaces, enhancing phagocytosis by making microbes more "sticky" for phagocytes.
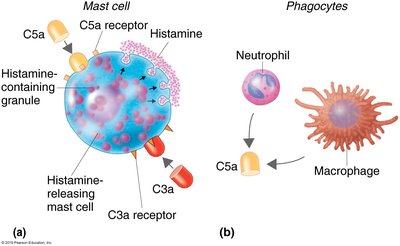
Inflammation: C3a and C5a bind to mast cells, causing the release of histamine and attracting phagocytes.
Interferons
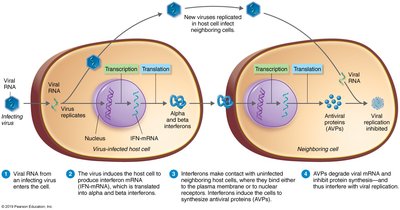
Interferons (IFNs) are cytokines with antiviral and antibacterial properties. They are produced by host cells in response to viral infections and help limit the spread of viruses.
IFN-α and IFN-β: Induce neighboring cells to produce antiviral proteins (AVPs) that inhibit viral replication.
IFN-γ: Activates neutrophils and macrophages to produce nitric oxide, which inhibits ATP production and kills bacteria and tumor cells.
Iron-Binding Proteins
Iron is essential for both host and microbial metabolism. The host uses iron-binding proteins to sequester iron and limit its availability to pathogens, thereby inhibiting their growth.
Transferrin: Found in blood and tissue fluids.
Lactoferrin: Present in milk, saliva, and mucus.
Ferritin: Stored in the liver, spleen, and bone marrow.
Hemoglobin: Located in red blood cells.
Bacteria may produce siderophores to scavenge iron from the host.

Antimicrobial Peptides (AMPs)
AMPs are small peptides produced by various cells and tissues as part of the innate immune response. They have broad-spectrum antimicrobial activity and are crucial for the first line of defense.
Produced by sweat glands (dermocidin), neutrophils (defensins), and other cells.
Mechanisms of action include inhibition of cell wall synthesis, formation of pores in microbial membranes, and destruction of DNA/RNA.
AMPs are effective against bacteria, viruses, fungi, and parasites, and microbes rarely develop resistance to them.
