 Back
BackMicrobial Metabolism: Fundamentals, Energy Conservation, and Catabolic Pathways
Study Guide - Smart Notes

Microbial Metabolism
Introduction to Metabolism
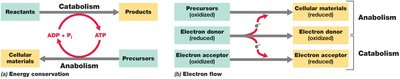
Metabolism encompasses all biochemical reactions required to sustain life in microorganisms. These reactions are divided into two main categories: catabolism (the breakdown of molecules to obtain energy) and anabolism (the synthesis of cellular materials using energy). Understanding metabolism is fundamental to predicting microbial behavior in diverse environments.
Catabolism: Exergonic reactions that release energy, often conserved as ATP.
Anabolism: Endergonic reactions that consume ATP to build cellular components.
Requirements for life: Liquid water, energy source, electron source, and nutrients for macromolecule synthesis.
Defining the Requirements for Life
All cells share fundamental metabolic requirements:
Water: Solvent for metabolic reactions.
Nutrients (including carbon): For synthesis of cellular materials.
Free energy: Energy available to do work, often stored as ATP.
Reducing power: The ability to donate electrons in metabolic reactions, essential for both catabolic and anabolic processes.
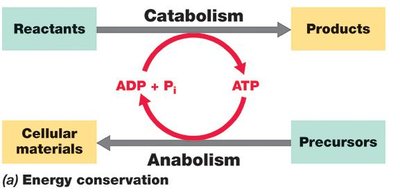
Cells conserve energy by converting it into ATP, which fuels cellular processes. The change in free energy during a reaction is denoted as (standard conditions: pH 7, 25°C, 1 atm, 1 M concentrations). Exergonic reactions () release energy, while endergonic reactions () require energy input.
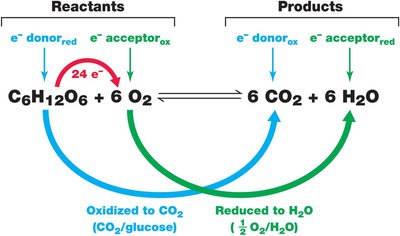
Example: Aerobic respiration of glucose: kJ/mol of glucose.
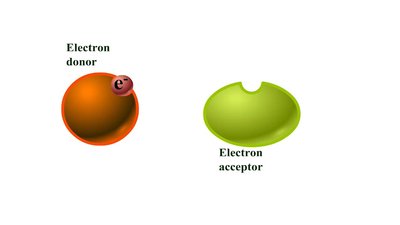
Reducing Power and Redox Reactions
Reducing power refers to the ability to donate electrons during redox (oxidation-reduction) reactions. These reactions involve an electron donor (oxidized) and an electron acceptor (reduced).
Electron donor: Transfers electrons and becomes oxidized.
Electron acceptor: Gains electrons and becomes reduced.
Example: In aerobic respiration of glucose, glucose is the electron donor and O2 is the electron acceptor.
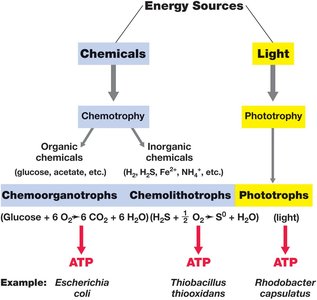
Metabolic Classes of Microorganisms
Microorganisms are classified based on their energy and carbon sources:
Phototrophs: Obtain energy from light (photosynthesis).
Chemotrophs: Obtain energy from chemical reactions.
Chemoorganotrophs: Use organic compounds for energy and electrons (e.g., sugars, proteins).
Chemolithotrophs: Use inorganic compounds for energy and electrons (e.g., H2, NH3, Fe2+).
Heterotrophs: Obtain carbon from organic compounds.
Autotrophs: Obtain carbon from CO2 (primary producers).
Electron Transfer Reactions
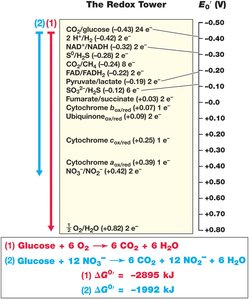
Redox Reactions and Reduction Potentials
Catabolism depends on electron flow from donors to acceptors. Redox reactions are characterized by reduction potential (), which measures a substance's affinity for electrons (in volts, relative to H2).
Redox couples are written as oxidized/reduced forms (e.g., NO3-/NO2-).
Negative : strong electron donors (e.g., glucose).
Positive : strong electron acceptors (e.g., O2).
The greater the difference in between donor and acceptor, the more energy is available.

Electron Carriers and N AD+/NADH Cycling
Electron transfer in cells is often mediated by soluble carriers such as nicotinamide adenine dinucleotide (NAD+/NADH). NADH is a good electron donor, while NAD+ is a weak acceptor. The reduction of NAD+ requires 2 electrons and 1 proton, producing NADH and releasing an extra proton into solution.
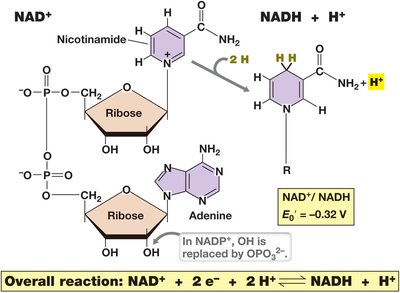
NAD+/NADH act as coenzymes, allowing various electron donors and acceptors to interact. NADP+/NADPH is used in anabolic reactions.
Cellular Energy Conservation
ATP and Energy-Rich Compounds
Adenosine triphosphate (ATP) is the primary energy currency in cells. It consists of adenosine and three phosphate groups, with two high-energy phosphate bonds. Hydrolysis of ATP to ADP or AMP releases significant free energy ( kJ/mol for ATP and ADP; kJ/mol for AMP).

Other energy-rich compounds include those with thioester bonds (e.g., acetyl-CoA). Cells require compounds with kJ/mol to synthesize ATP.
Mechanisms of Energy Conservation
Cells generate ATP through three main mechanisms:
Substrate-level phosphorylation: Direct transfer of a phosphate group from a phosphorylated compound to ADP (common in fermentation).
Oxidative phosphorylation: ATP synthesis driven by a proton motive force generated by electron transport (common in respiration).
Photophosphorylation: ATP synthesis using a proton motive force generated by light-driven electron transport (in phototrophs).
The proton motive force is an electrochemical gradient across the cytoplasmic membrane, essential for ATP synthesis in oxidative and photophosphorylation.
Catalysis and Enzymes
Enzyme Function and Activation Energy
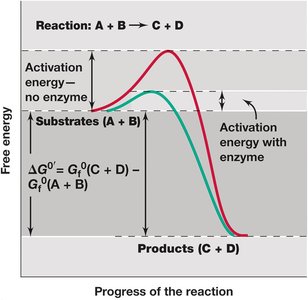
Free energy changes do not indicate reaction rates. Many reactions require an initial input of energy, called activation energy, to proceed. Catalysts (usually enzymes) lower the activation energy, increasing reaction rates without being consumed.

Enzyme Structure and Mechanism
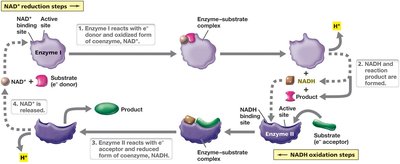
Enzymes are highly specific protein catalysts (some are RNA). They bind substrates at the active site, forming an enzyme-substrate complex, which is converted to product and released. The process is rapid and highly efficient.
Many enzymes require nonprotein molecules for activity:
Prosthetic groups: Tightly, often covalently bound (e.g., heme in cytochromes).
Coenzymes: Loosely, transiently bound (e.g., NADH), often derived from vitamins.
Enzyme catalysis involves proper substrate alignment and bond strain, reducing activation energy. Endergonic reactions are coupled to exergonic ones to ensure a negative or zero overall free energy change.
Catabolism: Chemoorganotrophs
Glycolysis (Embden–Meyerhof–Parnas Pathway)
Glycolysis is a nearly universal pathway for glucose catabolism, converting glucose to pyruvate. It consists of two stages:
Preparatory stage: Glucose is phosphorylated and split into two three-carbon intermediates.
Energy-conservation stage: Intermediates are oxidized, generating ATP and NADH, and forming pyruvate.
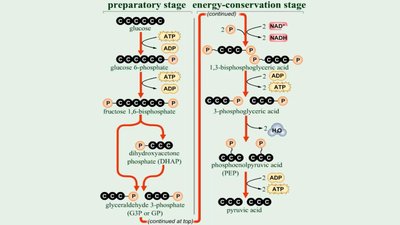
Key steps include phosphorylation, isomerization, cleavage, and ATP generation. Glycolysis uses 2 ATP in the preparatory stage and produces 4 ATP in the energy-conservation stage, yielding a net of 2 ATP, 2 NADH, and 2 pyruvate per glucose.
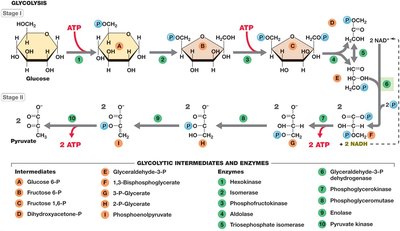
Glycolysis alone is insufficient for redox balance. Cells achieve redox balance by:
Fermentation: NADH is oxidized by reducing pyruvate to fermentation products.
Respiration: Pyruvate is further oxidized in the citric acid cycle, and electrons are transferred to external acceptors.
The Citric Acid Cycle (Krebs Cycle)
The citric acid cycle (CAC) oxidizes pyruvate to CO2, generating NADH, FADH2, and ATP. Acetyl-CoA (from pyruvate) combines with oxaloacetate to form citric acid, which is progressively oxidized, regenerating oxaloacetate.
Per pyruvate: 2 CO2, 3 NADH, 1 FADH2, 1 ATP (or GTP) produced.
Per glucose (2 pyruvate): 4 CO2, 6 NADH, 2 FADH2, 2 ATP.
The CAC also provides precursors for biosynthesis (e.g., amino acids, fatty acids, cytochromes).
Fermentation
Principles of Fermentation
Fermentation is an anaerobic process in which an organic compound serves as both electron donor and acceptor. ATP is produced by substrate-level phosphorylation, and redox balance is achieved by reducing pyruvate to waste products (e.g., ethanol, lactic acid).
Fermentation products are diverse and include ethanol, lactic acid, acetate, and other short-chain fatty acids. These products are important in food production and human health.
Alcohol fermentation (yeast): Glucose → 2 ethanol + 2 CO2
Lactic acid fermentation (bacteria): Glucose → 2 lactic acid
Fermentation supports a wide diversity of microorganisms but is less efficient than respiration in conserving energy.
Summary Table: Comparison of Catabolic Pathways
Pathway | Electron Acceptor | ATP Yield | Key Products |
|---|---|---|---|
Glycolysis | Internal (pyruvate or external in respiration) | 2 ATP (net) | Pyruvate, NADH |
Fermentation | Organic (pyruvate or derivative) | 2 ATP (from glycolysis) | Ethanol, lactic acid, other SCFAs |
Respiration | External (O2 or other) | ~38 ATP (aerobic, per glucose) | CO2, H2O |
Additional info: The above notes integrate and expand upon the provided lecture slides, images, and textbook references, ensuring a comprehensive and self-contained study guide for college-level microbiology students.
