 Back
BackMicrobial Metabolism II: Glycolysis, Citric Acid Cycle, and Microbial Metabolites
Study Guide - Smart Notes

Microbial Metabolism II
Introduction to Carbohydrate Catabolism
Carbohydrate catabolism is the process by which microbes break down complex sugars to release energy, primarily in the form of ATP. Microorganisms utilize a series of enzymatic reactions to convert large, non-transportable carbohydrates into smaller, absorbable molecules, which are then metabolized through central pathways such as glycolysis and the citric acid cycle.
Extracellular Digestion: Microbes secrete exoenzymes to degrade polymers like cellulose and starch outside the cell, producing smaller sugars that can be imported.
Uptake: Transport proteins (e.g., permeases) facilitate the import of monosaccharides and disaccharides into the cytoplasm for further metabolism.
Key Pathways: Glycolysis and the citric acid cycle are central to microbial energy production.
Glycolysis
Overview of Glycolysis
Glycolysis is a universal, anaerobic pathway that occurs in the cytoplasm of cells. It converts one molecule of glucose (6 carbons) into two molecules of pyruvate (3 carbons each), generating ATP and NADH in the process.
Location: Cytosol (cytoplasm)
Steps: 10 enzymatic reactions
Phases: Energy investment (uses ATP) and energy payoff (produces ATP and NADH)
Net Yield: 2 ATP, 2 NADH, 2 pyruvate per glucose
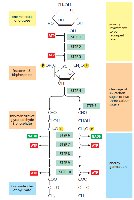
Stepwise Reactions of Glycolysis
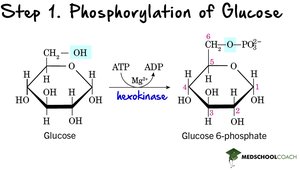
Step 1: Phosphorylation of Glucose – Glucose is phosphorylated by hexokinase/glucokinase using ATP, forming glucose-6-phosphate. This traps glucose inside the cell.
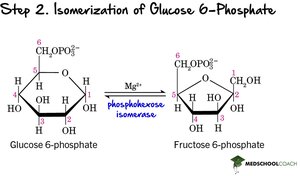
Step 2: Isomerization – Glucose-6-phosphate is converted to fructose-6-phosphate by phosphohexose isomerase.
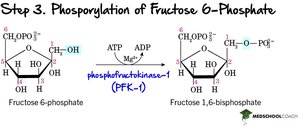
Step 3: Second Phosphorylation – Phosphofructokinase-1 (PFK-1) phosphorylates fructose-6-phosphate to fructose-1,6-bisphosphate (irreversible, regulatory step).
Step 4: Cleavage – Aldolase splits fructose-1,6-bisphosphate into two triose phosphates: glyceraldehyde-3-phosphate (G3P) and dihydroxyacetone phosphate (DHAP).
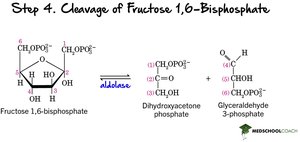
Step 5: Isomerization – Triose phosphate isomerase converts DHAP to G3P, so two G3P molecules proceed through the rest of glycolysis.
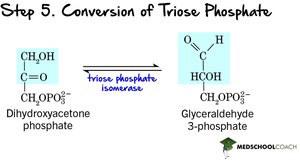
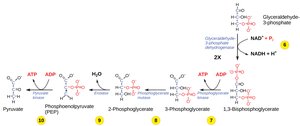
Step 6: Oxidation and Phosphorylation – G3P is oxidized, reducing NAD+ to NADH, and phosphorylated to 1,3-bisphosphoglycerate (no ATP used).
Step 7: Substrate-Level Phosphorylation – 1,3-bisphosphoglycerate donates a phosphate to ADP, forming ATP and 3-phosphoglycerate.
Step 8: Isomerization – 3-phosphoglycerate is converted to 2-phosphoglycerate by a mutase.
Step 9: Dehydration – Enolase removes water, forming phosphoenolpyruvate (PEP).
Step 10: Substrate-Level Phosphorylation – Pyruvate kinase transfers a phosphate from PEP to ADP, yielding ATP and pyruvate.
Summary Table: Glycolysis Steps and Key Enzymes
Step | Substrate | Product | Enzyme | ATP/NADH |
|---|---|---|---|---|
1 | Glucose | Glucose-6-phosphate | Hexokinase | -1 ATP |
2 | Glucose-6-phosphate | Fructose-6-phosphate | Phosphohexose isomerase | |
3 | Fructose-6-phosphate | Fructose-1,6-bisphosphate | PFK-1 | -1 ATP |
4 | Fructose-1,6-bisphosphate | G3P + DHAP | Aldolase | |
5 | DHAP | G3P | Triose phosphate isomerase | |
6 | G3P | 1,3-BPG | G3P dehydrogenase | +2 NADH |
7 | 1,3-BPG | 3-PG | Phosphoglycerate kinase | +2 ATP |
8 | 3-PG | 2-PG | Mutase | |
9 | 2-PG | PEP | Enolase | |
10 | PEP | Pyruvate | Pyruvate kinase | +2 ATP |
Fate of Pyruvate
Pyruvate, the end product of glycolysis, is a metabolic branch point. Its fate depends on oxygen availability and the organism's metabolic capabilities:
Aerobic Respiration: In the presence of oxygen, pyruvate is transported into mitochondria (eukaryotes) or remains in the cytoplasm (prokaryotes) and is converted to acetyl-CoA for entry into the citric acid cycle.
Fermentation: In the absence of oxygen, pyruvate is reduced to lactate (in animals) or ethanol (in yeast), regenerating NAD+ for glycolysis to continue.
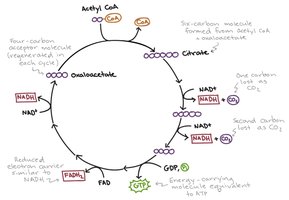
Citric Acid Cycle (Krebs Cycle)
Overview and Location
The citric acid cycle is a series of eight enzyme-catalyzed reactions that complete the oxidation of acetyl-CoA to CO2, generating NADH, FADH2, and ATP (or GTP). In eukaryotes, it occurs in the mitochondrial matrix; in prokaryotes, in the cytoplasm.
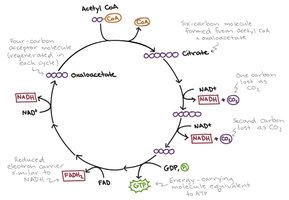
Stepwise Reactions of the Citric Acid Cycle
Step 1: Formation of Citrate – Acetyl-CoA combines with oxaloacetate to form citrate, catalyzed by citrate synthase.
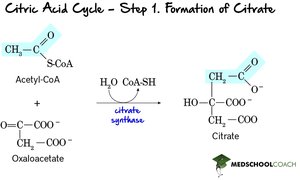
Step 2: Isomerization to Isocitrate – Citrate is rearranged to isocitrate via cis-aconitate intermediate, catalyzed by aconitase.
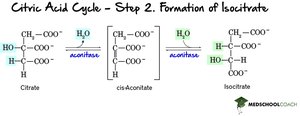
Step 3: Oxidative Decarboxylation of Isocitrate – Isocitrate is oxidized and decarboxylated to α-ketoglutarate, producing NADH and CO2.
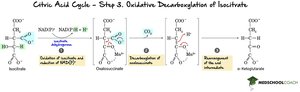
Step 4: Oxidative Decarboxylation of α-Ketoglutarate – α-Ketoglutarate is oxidized, releasing CO2 and forming succinyl-CoA, with NADH produced.
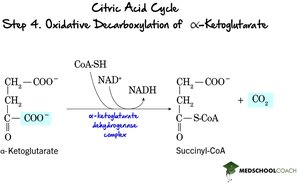
Step 5: Formation of Succinate – Succinyl-CoA is converted to succinate, generating ATP (or GTP) via substrate-level phosphorylation.
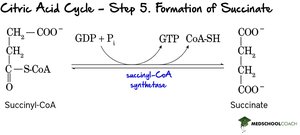
Step 6: Oxidation of Succinate – Succinate is oxidized to fumarate, reducing FAD to FADH2.
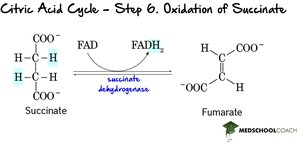
Step 7: Hydration of Fumarate – Fumarate is hydrated to malate.
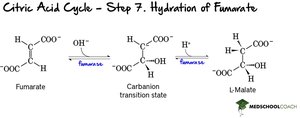
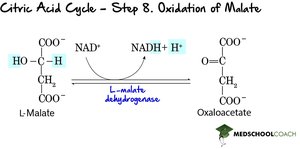
Step 8: Oxidation of Malate – Malate is oxidized to regenerate oxaloacetate, producing NADH.
Summary Table: Citric Acid Cycle Products (per Acetyl-CoA)
Product | Number Produced |
|---|---|
CO2 | 2 |
NADH | 3 |
FADH2 | 1 |
ATP (or GTP) | 1 |
Since one glucose yields two acetyl-CoA, the cycle turns twice per glucose molecule.
Microbial Metabolites and Host Interactions
Short-Chain Fatty Acids (SCFAs)
Gut microbes ferment dietary fiber to produce SCFAs such as acetate, propionate, and butyrate. These metabolites have significant effects on host physiology, including immune and nervous system modulation.
Immune System: Butyrate increases regulatory T cells (Tregs), which suppress excessive immune responses and inflammation.
Gut Barrier: SCFAs strengthen tight junctions between gut epithelial cells, preventing "leaky gut" and systemic inflammation.
Nervous System: SCFAs support microglia maturation and function, and stimulate the vagus nerve, influencing mood, appetite, and stress responses.
Neurotransmitter Production and Blood-Brain Barrier Protection
Microbial metabolism produces neuroactive compounds, influencing neurotransmitter levels and brain function.
Butyrate enhances the integrity of the blood-brain barrier, protecting the brain from pathogens and inflammatory molecules.
Tryptophan Metabolism
Microbes metabolize tryptophan into indoles, which interact with the aryl hydrocarbon receptor (Ahr) on immune cells. This signaling helps regulate immune responses and limits brain inflammation.
Key Equations
Glycolysis Net Reaction:
Citric Acid Cycle Net Reaction (per acetyl-CoA):
Additional info: The notes above expand on the original content by providing definitions, stepwise explanations, and tables for clarity. The role of microbial metabolites in host health is emphasized, reflecting current research in microbiome science.
