 Back
BackThe Human Microbiome: Structure, Function, and Clinical Relevance
Study Guide - Smart Notes

The Human Microbiome
Definition and Overview
The human microbiome refers to the vast collection of microorganisms—including bacteria, fungi, viruses, and archaea—and their genetic material that inhabit the human body. This 'second genome' contains up to 100 times more genes than the human genome and is essential for digestion, immunity, and disease prevention.

Key Body Sites: Microbial communities are highly specialized to unique environments such as the gut, skin, oral cavity, and vagina.
Genetic Diversity: The microbiome's gene pool vastly exceeds that of the human host, enabling a wide range of metabolic and protective functions.
Major Microbiome Niches
Gut Microbiome
The gut microbiome is the largest and most diverse microbial community in the body, dominated by the phyla Firmicutes and Bacteroidetes. It is crucial for breaking down complex carbohydrates, synthesizing vitamins, and training the immune system.
Metabolic Functions: Ferments indigestible dietary fibers into short-chain fatty acids (SCFAs) such as butyrate, propionate, and acetate.
Vitamin Synthesis: Produces essential vitamins, including Vitamin K and several B vitamins (B12, folate, biotin).
Immune Training: Approximately 70–80% of the immune system is located in the gut, where microbes help immune cells distinguish between harmless and harmful agents.
Critical Functions for Health
Metabolic Powerhouse
The gut microbiota provides metabolic services that the human body cannot perform independently. SCFAs produced by microbial fermentation serve as energy sources for colon cells and have anti-inflammatory and anticancer properties.
Vitamin Synthesis
Gut microbes synthesize essential vitamins, including Vitamin K (important for blood clotting) and B vitamins (such as B12, folate, and biotin).
Immune System Training
Microbes in the gut educate immune cells, helping them to tolerate harmless antigens and respond to pathogens. This process is vital for preventing autoimmune and allergic diseases.
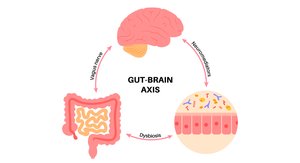
The Gut-Brain Axis
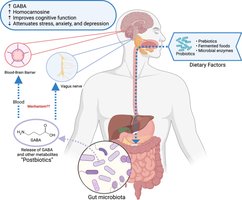
The gut-brain axis is a bidirectional communication network linking the gut microbiota and the brain via the vagus nerve and microbial metabolites. Microbes produce neurotransmitters such as serotonin and GABA, influencing mood, stress, and cognition.
Diet, Lifestyle, and the Microbiome
Dietary Fiber and Microbial Health
A diet rich in diverse plant fibers (fruits, vegetables, legumes, whole grains) is the primary fuel for beneficial gut bacteria. Insufficient fiber can lead microbes to degrade the gut's protective mucus layer, increasing the risk of 'leaky gut.'


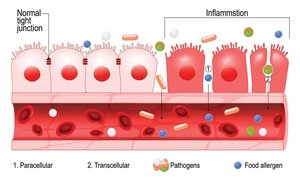
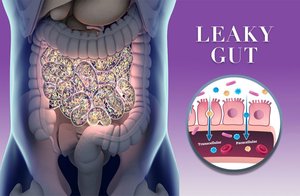
Leaky Gut (Increased Intestinal Permeability)
Leaky gut refers to increased intestinal permeability, where the gut lining becomes more porous, allowing bacteria, toxins, and undigested food particles to enter the bloodstream. This can trigger chronic systemic inflammation.
Dietary Risks: High Fat and Sugar
Diets high in sugar, saturated fats, and ultra-processed foods reduce microbial diversity and promote pro-inflammatory species, increasing the risk of obesity and metabolic disease.

Antibiotics and Microbial Disruption
Antibiotics, while life-saving, can disrupt the microbiome by eliminating beneficial bacteria and allowing opportunistic pathogens (e.g., Clostridioides difficile) to proliferate.

The Microbiota-Gut-Brain Axis
Microbial Communication with the Brain
Gut bacteria communicate with the brain through the microbiota-gut-brain axis, influencing mood, stress responses, and cognitive function. Transplanting gut bacteria from humans with depression into mice can induce depressive-like behaviors in the animals.
Neurotransmitter Production
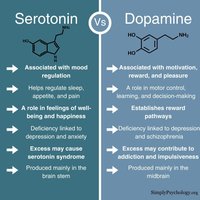
Serotonin: 90–95% of the body's serotonin is produced in the gut. Bacteria such as Lactobacillus and Bifidobacterium regulate tryptophan, the precursor to serotonin.
GABA: The primary inhibitory neurotransmitter, synthesized by bacteria like Bifidobacterium dentium and Lactobacillus rhamnosus, helps calm the nervous system and reduce anxiety.
Dopamine: About 50% of the body's dopamine is synthesized in the GI tract by species such as Staphylococcus and Enterococcus.

Therapeutic Psychobiotics
Definition and Mechanisms
Psychobiotics are a class of probiotics and prebiotics specifically used to treat or manage mental health disorders and improve cognitive function. They communicate with the brain through neural, endocrine, and metabolic pathways.

Neural Pathway: Signals are transmitted via the vagus nerve, directly connecting the gut and brain.
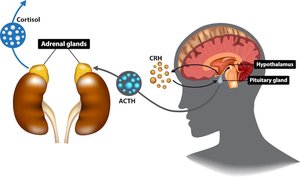
Endocrine Pathway: Psychobiotics regulate the HPA axis, lowering cortisol and other stress hormones.
Metabolic Pathway: They stimulate the production of neurotransmitters and SCFAs that can cross the blood-brain barrier.
Therapeutic Applications
Depression and anxiety
Stress management
Neurodegenerative diseases
Neurodevelopmental disorders
Microbiome Acquisition and the Hygiene Hypothesis
Hygiene and Microbiome Hypotheses
The hygiene hypothesis suggests that reduced exposure to microbes in early childhood increases the risk of allergic and autoimmune diseases. The microbiome hypothesis extends this idea, emphasizing the importance of acquiring a diverse community of beneficial microbes early in life for proper immune system training.
Key Drivers of Microbiome Acquisition
Birth Method: Vaginal delivery provides the first major microbial inoculation, while C-sections are linked to different initial gut compositions and higher asthma risk.
Infant Feeding: Breast milk contains beneficial bacteria and oligosaccharides that feed healthy gut microbes like Bifidobacterium infantis.
Environmental Exposure: Growing up on farms, having pets, or living with older siblings increases exposure to diverse microbes, lowering allergy rates.


Immunological Mechanism
Microbes stimulate regulatory T cells (Tregs), which prevent the immune system from overreacting to harmless substances. Without these signals, the immune system may become hypersensitive, leading to allergies.

Benefits and Risks of the Microbiota
Core Benefits of a Healthy Microbiota
Immune Training: Reduces risk of allergies and asthma by educating immune cells.
Nutrient Synthesis: Sole producers of Vitamin B12, Vitamin K, folate, and essential amino acids.
Metabolic Support: Breaks down fiber into SCFAs, providing energy and regulating appetite and blood sugar.
Pathogen Protection: Provides colonization resistance, crowding out harmful bacteria like C. difficile.
Risks Associated with Dysbiosis
Dysbiosis is a disrupted microbial balance, often caused by antibiotics, poor diet, or chronic stress. It is linked to:
Chronic inflammation and 'leaky gut'
Autoimmune and digestive disorders (e.g., IBD, rheumatoid arthritis, lupus)
Metabolic issues (e.g., obesity, diabetes)
Mental health impacts (e.g., anxiety, depression)
Therapeutic Risks
Interventions such as Fecal Microbiota Transplants (FMT) can restore balance but may also transfer microbes that alter metabolism or behavior.

Microbiota and Metabolic Health
Microbiota and Leanness
No single microbe guarantees weight loss, but a diverse and balanced gut microbiota supports metabolic health by improving energy regulation, influencing fat storage, modulating inflammation, and interacting with appetite and glucose metabolism hormones.

Lower Energy Harvest: Lean microbiota may extract fewer calories from food, allowing more energy to pass through the system.
Increased Satiety: SCFAs like propionate and butyrate trigger hormones (GLP-1 and PYY) that signal fullness to the brain.
Metabolic Boosting: Certain microbes (e.g., Christensenella minuta) increase host energy expenditure and physical activity.
Reduced Fat Storage: Some bacteria inhibit enzymes (like LPL) responsible for fat storage, directing energy toward use rather than storage.
