 Back
BackFundamental Chemistry Concepts for Nutrition Students
Study Guide - Smart Notes

What Is Matter?
Definition and States of Matter
Matter is defined as anything that has mass and occupies space. It exists in four states: solid, liquid, gas, and plasma. All matter, whether living or nonliving, is composed of elements.
Element: A substance that cannot be broken down by chemical means into other substances.
Examples: Fish, cats, people, soil, desk.
95% of the body weight of organisms is composed of six elements: Carbon (C), Hydrogen (H), Oxygen (O), Nitrogen (N), Phosphorus (P), Sulfur (S) (acronym: CHONPS).

Elements and the Periodic Table
Organization of Elements
The periodic table organizes elements by their atomic number and chemical properties. Elements are arranged horizontally in rows (periods) and vertically in columns (groups).
Atoms in the same group share similar chemical binding characteristics.
Group VIII (noble gases) are inert and do not react chemically.
Atoms shown in the periodic table are electrically neutral.
Atomic Number and Mass Number
Each element is represented by a unique atomic symbol (e.g., H for hydrogen, Na for sodium).
Atomic number: Number of protons (and electrons) in an atom.
Mass number: Sum of protons and neutrons in the nucleus.
Atomic mass: Approximately equal to the mass number.
Atoms: Structure and Properties
Subatomic Particles
An atom is the smallest part of an element that displays its properties. Atoms consist of a central nucleus (containing protons and neutrons) and orbiting electrons.
Protons: Positively charged.
Neutrons: No charge.
Electrons: Negatively charged, very low mass.
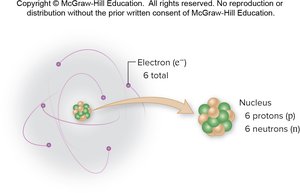
Electron Shells and Valence Electrons
Electrons occupy shells around the nucleus. The distribution of electrons determines the chemical behavior of atoms.
First shell holds up to 2 electrons; subsequent shells hold up to 8.
Valence shell: The outermost shell, determines chemical properties.
Octet rule: Atoms are most stable when their valence shell has 8 electrons (or 2 for atoms with only one shell).
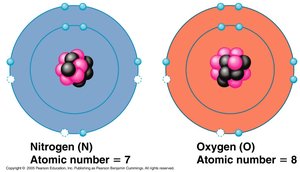
Isotopes
Definition and Applications
Isotopes are atoms of the same element with different numbers of neutrons, resulting in different atomic masses. Some isotopes are radioactive and decay spontaneously, emitting energy.
Used as tracers in medicine (e.g., I-131 for thyroid scans).
Examples: Carbon-12, Carbon-13, Carbon-14.
Molecules and Compounds
Formation and Chemical Formulas
Atoms form chemical bonds to fill their valence shells, resulting in molecules (same atoms) or compounds (different atoms).
Noble gases are inert because their valence shells are full.
A chemical formula indicates the number and type of atoms in a molecule (e.g., C6H12O6 for glucose).

Electronegativity and Chemical Bonds
Electronegativity
Electronegativity measures an atom's ability to attract electrons. Differences in electronegativity determine the type of chemical bond formed between atoms.
High electronegativity: Strong attraction for electrons.
Low electronegativity: Weak attraction for electrons.
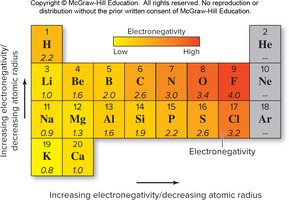
Types of Chemical Bonds
Covalent bonds: Atoms share electrons. Can be nonpolar (equal sharing) or polar (unequal sharing).
Nonpolar covalent bonds: Electrons are shared equally (e.g., hydrogen gas, oxygen gas, methane).

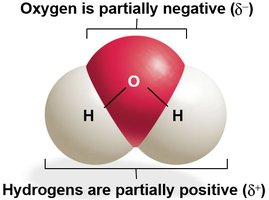
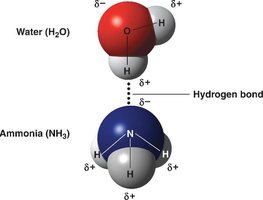
Polar covalent bonds: Electrons are shared unequally, resulting in partial charges (e.g., water molecule).
Ionic bonds: Electrons are transferred from one atom to another, forming oppositely charged ions (e.g., NaCl).
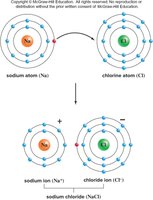
Hydrogen bonds: Weak bonds between polar covalent compounds, important for structure and function of water, proteins, and DNA.
Chemistry and Properties of Water
Unique Properties of Water
Water is essential for life, making up 70-90% of living organisms. Its molecules cling together due to hydrogen bonding, giving water unique properties.
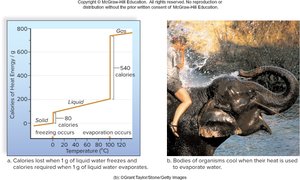
High heat capacity: Water absorbs large amounts of heat with minimal temperature change.
High heat of evaporation: Organisms cool as water evaporates from their bodies.
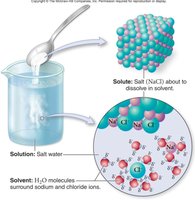
Water as a Solvent
Water's polarity allows it to dissolve many substances, making it a universal solvent. Hydrophilic substances (polar molecules/ions) dissolve readily in water.
Solution: Contains dissolved substances (solutes) and solvent (water).
Cohesion and Adhesion
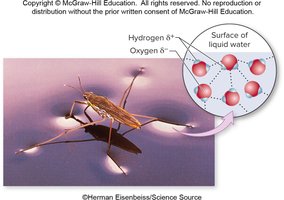
Water molecules exhibit cohesion (cling to each other) and adhesion (cling to other polar surfaces), both due to hydrogen bonding and polarity.
Cohesion: Contributes to surface tension, allowing insects to walk on water.
Adhesion: Enables water to move through plant vessels and adhere to surfaces.

Density and Insulation
Water is less dense as a solid than as a liquid. Below 4°C, hydrogen bonds become more rigid and open, causing ice to float and act as an insulator.
Ice floating prevents oceans from freezing solid.
Ice insulates aquatic life during winter.

Water in Chemical Reactions
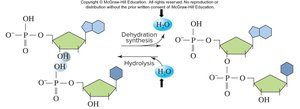
Water participates in essential chemical reactions, including those that build and break down biological molecules.
Dehydration synthesis: Water is released when molecules are built.
Hydrolysis: Water is used to break down molecules.
Acids, Bases, and pH
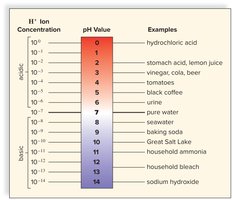
pH Scale
The pH scale measures the acidity or basicity of a solution, ranging from 0 (acidic) to 14 (basic). It is a logarithmic scale, where each unit represents a tenfold change in hydrogen ion concentration.
pH < 7: Acidic
pH = 7: Neutral
pH > 7: Basic (alkaline)
Example: pH 4 is 10× more acidic than pH 5.
Buffers and Homeostasis
Buffers are chemicals that help maintain pH within normal limits, essential for homeostasis in organisms. Human blood is normally pH 7.4; deviations can cause acidosis or alkalosis.
Example: Carbonic acid buffer system in blood.
Summary Table: Types of Chemical Bonds
Bond Type | Mechanism | Strength | Example |
|---|---|---|---|
Covalent (Nonpolar) | Equal sharing of electrons | Strong | H2, O2, CH4 |
Covalent (Polar) | Unequal sharing of electrons | Strong | H2O |
Ionic | Transfer of electrons | Strong | NaCl |
Hydrogen | Attraction between polar molecules | Weak (collectively strong) | Water, DNA |
