 Back
BackIntroduction to Biomolecules and Chemistry in Nutrition
Study Guide - Smart Notes

Science of Nutrition
Physiological Processes Regulated by Chemistry
Nutrition is fundamentally linked to chemistry, as physiological processes in the human body rely on chemical reactions and biomolecules. These processes include digestion, metabolism, oxygen transport, neuronal signaling, cell communication, muscle contraction, and hormone synthesis.
Digestion and Metabolism: Chemical reactions break down food into nutrients and energy.
Oxygen Uptake and Delivery: Hemoglobin and other molecules facilitate oxygen transport.
Neuronal Signaling: Neurotransmitters and ions are involved in nerve impulse transmission.
Cell-Cell Communication: Hormones and signaling molecules regulate cellular activities.
Fundamental Units of Matter
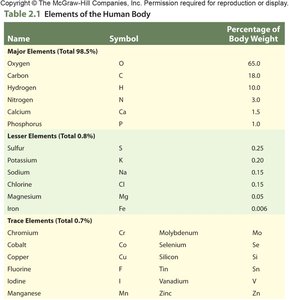
Elements of the Human Body
The human body is composed primarily of six elements, which make up 98.5% of its mass. These elements are essential for life and are involved in various biochemical processes.
Major Elements: Oxygen, Carbon, Hydrogen, Nitrogen, Calcium, Phosphorus
Lesser Elements: Sulfur, Potassium, Sodium, Chlorine, Magnesium
Trace Elements: Chromium, Cobalt, Copper, Fluorine, Manganese, Molybdenum, Selenium, Silicon, Tin, Vanadium, Zinc
Name | Symbol | Percentage of Body Weight |
|---|---|---|
Oxygen | O | 65.0 |
Carbon | C | 18.0 |
Hydrogen | H | 10.0 |
Nitrogen | N | 3.0 |
Calcium | Ca | 1.5 |
Phosphorus | P | 1.0 |
Sulfur | S | 0.25 |
Potassium | K | 0.20 |
Sodium | Na | 0.15 |
Chlorine | Cl | 0.15 |
Magnesium | Mg | 0.05 |
Trace Elements | - | 0.7 |
Atoms, Molecules, and Compounds
Basic Chemical Concepts
Atoms are the smallest units of elements and serve as the building blocks of molecules. Molecules are composed of two or more atoms, while compounds are molecules made from two or more different elements.
Atom: Smallest unit of an element.
Molecule: Two or more atoms bonded together.
Compound: Molecule with atoms from different elements.

The Human Body & Digestion
Atomic Structure and Chemical Bonds
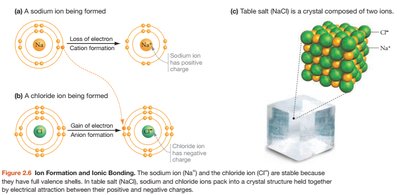
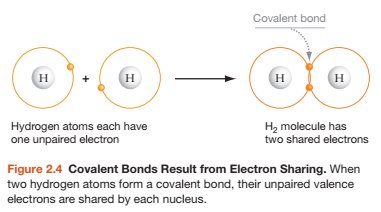
Atoms interact through chemical bonds, which are essential for forming molecules and compounds. The main types of chemical bonds are ionic, covalent, and hydrogen bonds.
Ionic Bonds: Formed when one atom transfers electrons to another, resulting in charged ions that attract each other. Example: NaCl (table salt).
Covalent Bonds: Formed when atoms share electrons. Can be polar (unequal sharing) or non-polar (equal sharing).
Hydrogen Bonds: Weak bonds between a δ+ and δ− charge, important in water and biological molecules.

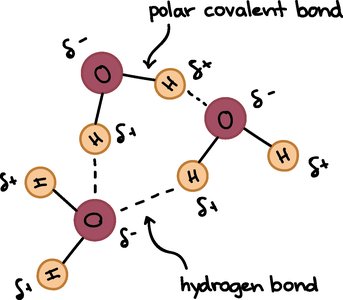
Essential Chemicals for Life
Organic and Inorganic Compounds
Organic compounds contain carbon and are typically covalently bonded, while inorganic compounds lack carbon and are often simpler. Water is the most abundant inorganic compound in the body and is crucial for dissolving chemicals and maintaining physiological functions.
Organic Compounds: Examples include glucose (C6H12O6).
Inorganic Compounds: Examples include water (H2O), salts (NaCl, KCl).
Water: Excellent solvent, liquid at a wide range of temperatures.
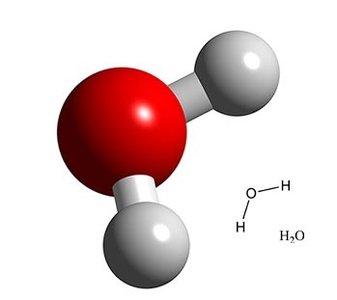

Water and Minerals
Properties of Water
Water's polarity and hydrogen bonding make it an excellent solvent for polar molecules and ionic compounds. Nonpolar molecules do not dissolve in water, which is important for biological separation of substances.
Solvent Properties: Water dissolves salts and sugars, but not fats.
Hydrogen Bonding: Responsible for water's unique properties, such as high heat capacity and surface tension.
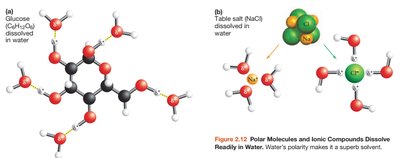
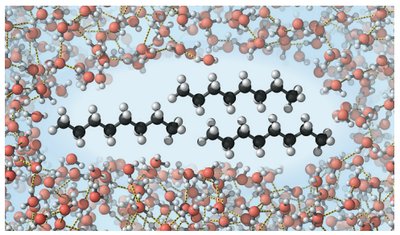
Organic Compounds
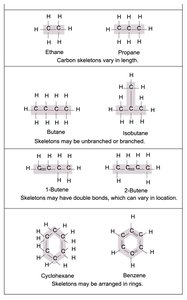
Hydrocarbons and Functional Groups
Hydrocarbons are composed of carbon and hydrogen and are nonpolar. Isomers have the same atoms but different structural arrangements. Functional groups determine the chemical properties and reactivity of organic molecules.
Hydrocarbons: Basis for many biomolecules.
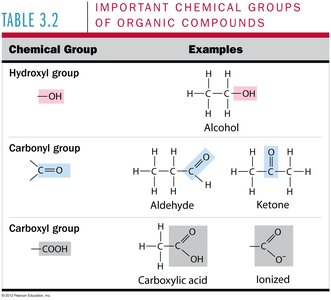
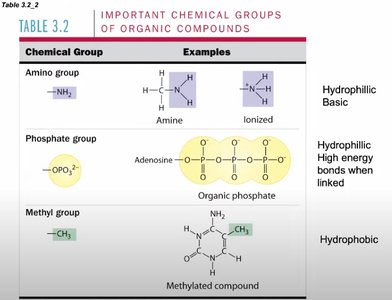
Functional Groups: Include hydroxyl, carbonyl, carboxyl, amino, phosphate, and methyl groups.
Important Biochemicals
Classes of Biomolecules
There are four major classes of biomolecules essential for life: carbohydrates, proteins, lipids, and nucleic acids. Each class has unique structures and functions in the body.
Carbohydrates: Sugars and starches, provide energy.
Proteins: Contain nitrogen, serve as enzymes, hormones, and structural components.
Lipids: Fats, energy storage, cell membranes.
Nucleic Acids: DNA and RNA, genetic information.
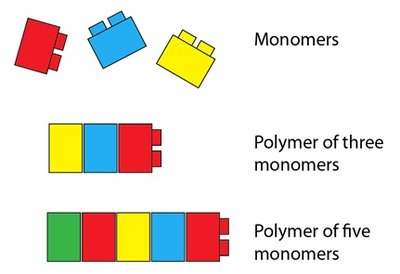
Monomers and Polymers
Building Blocks of Biomolecules
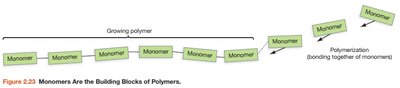
Monomers are small molecules that can be joined end-to-end to form polymers. Polymers are large molecules made from repeating monomer units. Examples include proteins (amino acids), carbohydrates (monosaccharides), and nucleic acids (nucleotides).
Monomer: Small molecule, e.g., amino acid, monosaccharide.
Polymer: Large molecule, e.g., protein, polysaccharide.
Polymerization: Process of joining monomers.
Synthesizing and Breaking Down Polymers
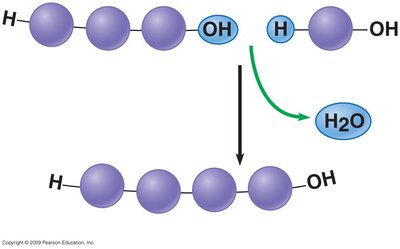
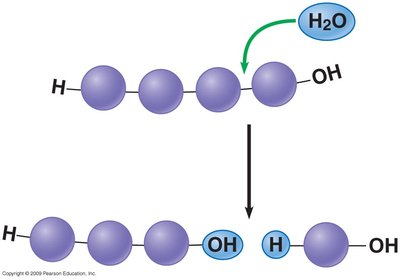
Polymers are synthesized by dehydration synthesis, where water is removed to join monomers. They are broken down by hydrolysis, where water is added to split polymers into monomers.
Dehydration Synthesis: Joins monomers, releases water.
Hydrolysis: Breaks polymers, consumes water.
Lipids
Structure and Function
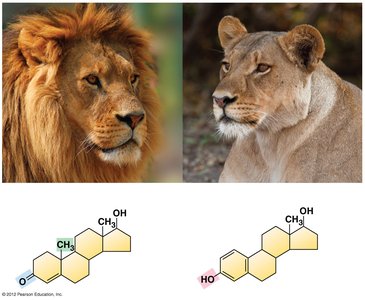
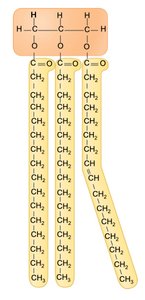
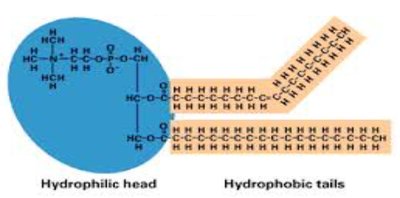
Lipids are hydrophobic molecules composed mostly of carbon and hydrogen. They include fatty acids, triglycerides, phospholipids, and steroids, each with distinct roles in energy storage, cell membranes, and hormone synthesis.
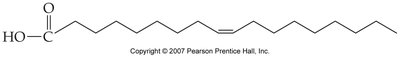
Fatty Acids: Long hydrocarbon chains with a carboxylic acid group.
Triglycerides: Glycerol linked to three fatty acids, main energy storage form.
Phospholipids: Glycerol, two fatty acids, and a phosphate group; major component of cell membranes.
Steroids: Four carbon rings; function as hormones and membrane components.

Carbohydrates
Structure and Function
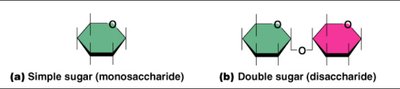
Carbohydrates are composed of carbon, hydrogen, and oxygen. Their monomers are monosaccharides (simple sugars), which can be linked to form polysaccharides (complex carbohydrates).
Monosaccharides: Simple sugars, e.g., glucose.
Polysaccharides: Chains of monosaccharides, e.g., starch, glycogen.
Proteins
Structure and Function
Proteins are polymers of amino acids, which contain carbon, hydrogen, oxygen, and nitrogen. The sequence and shape of proteins determine their function, including acting as enzymes, hormones, and antibodies.
Amino Acids: Monomers of proteins.
Protein Function: Enzymes, structural support, signaling.
Shape Determines Function: Protein folding is critical for activity.
Enzymes
Biological Catalysts
Enzymes are proteins that increase the rate of chemical reactions in the body. They are essential for digestion and cellular metabolism. Factors that alter enzyme shape can affect their function.
Catalysis: Enzymes lower activation energy for reactions.
Specificity: Each enzyme acts on a specific substrate.
Nucleic Acids
DNA and RNA
Nucleic acids are polymers made from nucleotide monomers. DNA stores genetic information, while RNA is involved in protein synthesis.
DNA: Deoxyribonucleic acid, genetic material.
RNA: Ribonucleic acid, helps translate DNA into proteins.
Energy Currency of the Cell
Adenosine Triphosphate (ATP)
ATP is the primary energy carrier in cells. Energy is released by breaking high-energy phosphate bonds, and ATP is replenished by the oxidation of food fuels.
ATP Structure: Adenosine molecule with three phosphate groups.
Energy Release: Hydrolysis of ATP releases energy for cellular processes.
Summary Table: Major Biomolecules
Class | Monomer | Function |
|---|---|---|
Carbohydrates | Monosaccharide | Energy, structure |
Proteins | Amino acid | Enzymes, structure, signaling |
Lipids | Fatty acid | Energy storage, membranes |
Nucleic Acids | Nucleotide | Genetic information |
Additional info: Academic context was added to clarify the role of biomolecules in nutrition and physiological processes, and to ensure completeness for exam preparation.
