 Back
BackProteins and Amino Acids: Structure, Function, and Digestion
Study Guide - Smart Notes

Overview of Proteins
Definition and Importance
Proteins are large, complex molecules composed of one or more chains of amino acids linked by peptide bonds. Their unique three-dimensional structures enable them to perform a vast array of functions essential for life, including structural support, catalysis, transport, and regulation.
Structural support: Proteins like collagen provide strength to bones and connective tissues.
Muscle contraction: Actin and myosin are key proteins in muscle fibers.
Transport: Hemoglobin transports oxygen in the blood.
Hormone regulation: Insulin and glucagon regulate blood glucose levels.
Immune response: Antibodies defend against pathogens.
Cell signaling: Receptors transmit signals across cell membranes.
Ion transport: Ion channels maintain cellular homeostasis.
Enzymatic activity: Enzymes catalyze biochemical reactions.
Distribution in the Body
40% of body protein is found in skeletal muscle.
25% is within organs.
35% is distributed throughout the skin and blood.
Amino Acids
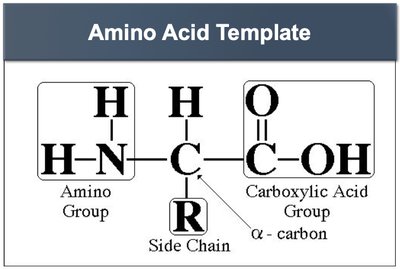
Structure of Amino Acids
Amino acids are the building blocks of proteins. Each amino acid contains a central carbon atom (α-carbon) bonded to a hydrogen atom, an amino group (-NH2), a carboxyl group (-COOH), and a variable side chain (R group) that determines its unique properties.
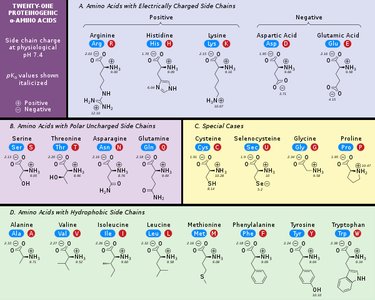
There are 20 standard amino acids found in proteins.
The sequence and chemical properties of amino acids dictate protein structure and function.
Categorization of Amino Acids
Amino acids can be grouped by their chemical structure or by their essentiality in the diet.
By structure: Grouped based on similarities in their side chains (e.g., hydrophobic, hydrophilic, acidic, basic).
By essentiality: Classified as non-essential, conditionally essential, or essential.
Essentiality of Amino Acids
Non-essential amino acids: Can be synthesized by the body (e.g., alanine, aspartic acid, asparagine, glutamic acid, glycine, serine).
Conditionally essential amino acids: Normally synthesized by the body, but may be required from the diet under certain conditions (e.g., arginine, cysteine, glutamine, proline, tyrosine).
Essential amino acids: Cannot be synthesized by the body and must be obtained from the diet. There are nine: histidine, isoleucine, leucine, lysine, methionine, phenylalanine, threonine, tryptophan, and valine.

Mnemonic for Essential Amino Acids
"I'm Very Tall, Have Long Legs, & Play Music Tenderly" helps recall: Isoleucine, Valine, Threonine, Histidine, Leucine, Lysine, Phenylalanine, Methionine, Tryptophan.

Functions and Food Sources of Essential Amino Acids
Isoleucine: Muscle metabolism, immune function, energy production. Sources: chicken breast, soybeans, turkey, lean beef.
Valine: Muscle metabolism, tissue repair, nitrogen balance. Sources: soybeans, lean beef, chicken, salmon.
Threonine: Collagen formation, immune function, nitrogen balance. Sources: chicken, turkey, salmon, lentils.
Histidine: Synthesis of histamine, pH balance. Sources: parmesan cheese, pork loin, turkey, tuna.
Leucine: Muscle protein synthesis, energy production, blood sugar regulation. Sources: beef, chicken, tuna, soybeans.
Lysine: Protein synthesis, tissue repair, calcium absorption. Sources: lean beef, chicken, salmon, eggs.
Phenylalanine: Precursor for neurotransmitters, protein synthesis. Sources: lean beef, chicken, fish, eggs.
Methionine: Protein synthesis, methylation, antioxidation. Sources: beef, chicken, fish, eggs.
Tryptophan: Precursor for serotonin, melatonin, niacin. Sources: turkey, chicken, pumpkin seeds, soybeans, dairy.
Branched-Chain Amino Acids (BCAAs)
BCAAs include leucine, isoleucine, and valine. They are characterized by their branched molecular structure and play crucial roles in muscle protein synthesis, metabolism, and energy production. BCAAs are especially important for muscle recovery and may help reduce exercise-induced muscle damage.

Protein Structure
Levels of Protein Structure
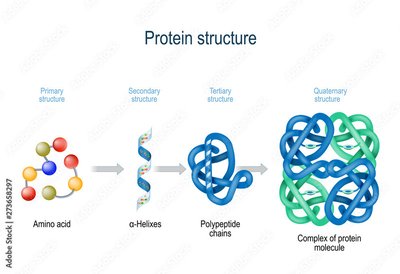
The function of a protein is determined by its structure, which is organized into four hierarchical levels:
Primary structure: Linear sequence of amino acids determined by the genetic code. Peptide bonds link amino acids together.
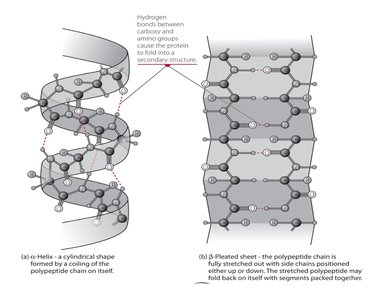
Secondary structure: Local folding into alpha helices and beta sheets, stabilized by hydrogen bonds.
Tertiary structure: The overall three-dimensional shape formed by interactions among side chains, including hydrogen bonds and disulfide bridges.
Quaternary structure: The arrangement of multiple polypeptide chains into a functional protein complex.


Protein Digestion
Overview of Digestion
Protein digestion is a multi-step process that begins in the stomach and is completed in the small intestine. The goal is to break down dietary proteins into absorbable amino acids and small peptides.
Mouth
Mechanical digestion begins as food is chewed.
No enzymatic digestion of proteins occurs in the mouth.
Stomach
Gastric cells release gastrin in response to protein, stimulating secretion of hydrochloric acid (HCl).
HCl denatures protein structures, making them accessible to enzymes.
HCl activates pepsinogen to pepsin, which hydrolyzes peptide bonds, producing large polypeptides and free amino acids.
Small Intestine
Acidic chyme stimulates release of secretin and cholecystokinin (CCK).
Secretin triggers pancreatic secretion of bicarbonate and protease zymogens (trypsinogen, chymotrypsinogen, procarboxypeptidases).
These zymogens are activated to trypsin, chymotrypsin, and carboxypeptidase, which further digest proteins.
Brush border peptidases break oligopeptides into dipeptides, tripeptides, and free amino acids for absorption.
Summary Table: Key Concepts
Concept | Description |
|---|---|
Proteins | Large, complex molecules made of amino acids; essential for structure, function, and regulation of tissues and organs. |
Amino Acids | Building blocks of proteins; 20 standard types with unique side chains. |
Non-essential AA | Can be synthesized by the body. |
Conditionally Essential AA | Normally synthesized by the body, but may be required from diet under certain conditions. |
Essential AA | Cannot be synthesized by the body; must be obtained from diet. |
Primary Structure | Linear sequence of amino acids. |
Secondary Structure | Alpha helices and beta sheets formed by hydrogen bonding. |
Tertiary Structure | Three-dimensional folding due to side chain interactions. |
Protein Digestion | Stomach: HCl and pepsin; Small intestine: pancreatic proteases and brush border enzymes. |
