 Back
BackProteins: Structure, Function, Digestion, and Dietary Guidelines
Study Guide - Smart Notes

Proteins: Structure, Function, Digestion, and Dietary Guidelines
Introduction to Proteins
Proteins are a functionally diverse class of macromolecules essential for life. They are composed of one or more chains of amino acids, typically ranging from 50 to 10,000 amino acids joined by peptide bonds. Unlike carbohydrates and lipids, proteins are a significant source of nitrogen in the diet. Common dietary sources include meat, poultry, seafood, dairy products, whole grains, legumes, and nuts/seeds.

Definition: Proteins are polymers of amino acids linked by peptide bonds.
Functions: Building new proteins, synthesizing other nitrogen-containing molecules, and providing energy.
Unique Feature: Proteins are the only macronutrient that contains nitrogen.
Amino Acid Structure
Proteins are built from 20 different amino acids, each sharing a basic structure but differing in their side chain (R group), which gives each amino acid unique properties. Nine of these amino acids are essential and must be obtained from the diet, while the remaining eleven can be synthesized by the body.
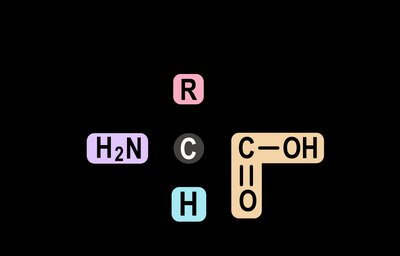
Key Components: Central carbon (C), amino group (H2N), carboxyl group (COOH), hydrogen atom, and variable R group.
Essential Amino Acids: Must be consumed in the diet.
Nonessential Amino Acids: Can be synthesized by the body.
Levels of Protein Structure
The structure of a protein determines its function. Proteins have four levels of structure:

Primary Structure: The sequence of amino acids in a polypeptide chain.
Secondary Structure: Local folding into α-helices and β-sheets due to hydrogen bonding.
Tertiary Structure: The overall three-dimensional shape of a single polypeptide chain, determined by interactions among R groups.
Quaternary Structure: Association of multiple polypeptide chains to form a functional protein.
Example: Sickle cell anemia is caused by a single amino acid change in the primary structure of hemoglobin, affecting all higher levels of structure and function.
Protein Denaturation
Denaturation is the process by which a protein loses its native structure and, consequently, its function. This can be caused by heat, acids/bases, mechanical agitation, or salts. Denaturation affects all levels of protein structure except the primary structure.

Causes: Heat (e.g., cooking an egg), acids/bases (e.g., marinating meat in lemon juice), mechanical agitation (e.g., whipping egg whites), and salts.
Effect: Loss of biological function.




Protein Synthesis: Gene Expression
Protein synthesis is governed by gene expression, which involves two main steps: transcription and translation. Genes (segments of DNA) are transcribed into messenger RNA (mRNA), which is then translated by ribosomes into a specific sequence of amino acids, forming a protein.
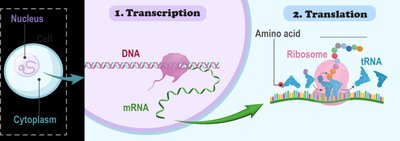
Transcription: DNA → mRNA
Translation: mRNA → Amino acid chain (protein)
Ribosomes: Facilitate the linking of amino acids via peptide bonds during translation.
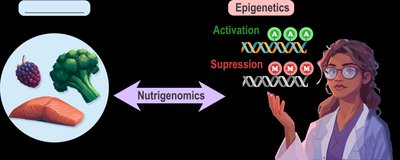
Epigenetics refers to chemical modifications that affect gene expression without altering the DNA sequence. Nutrigenomics studies how dietary components influence gene expression, impacting health and disease risk.
Functions of Proteins in the Body
Proteins are required for numerous physiological functions and must constitute 10–35% of total daily energy intake. They are essential for:
Biological Compounds: Enzymes, hormones, transport proteins, neurotransmitters, and blood clotting factors.
Immunity: Antibodies are proteins that neutralize pathogens.
Structural and Mechanical Support: Collagen (skin, bones), actin, and myosin (muscle contraction).
Growth and Repair: Required throughout the lifespan for tissue growth and repair.
Balance: Fluid, electrolyte, and acid-base balance (proteins act as buffers).
Energy: Backup energy source via deamination (4 kcal/g).
Protein Digestion and Absorption
Protein digestion begins in the stomach and is completed in the small intestine. The process involves denaturation by stomach acid and enzymatic cleavage by proteases, resulting in single amino acids that are absorbed into the bloodstream.
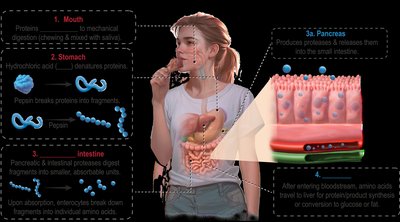
Step | Location | Enzyme/Agent | Product |
|---|---|---|---|
1 | Mouth | N/A (mechanical digestion) | Chewed proteins |
2 | Stomach | HCl, Pepsin | Denatured proteins, peptide fragments |
3 | Small Intestine | Pancreatic & intestinal proteases | Dipeptides, tripeptides, amino acids |
4 | Enterocytes (intestinal cells) | Dipeptidases, tripeptidases | Single amino acids |
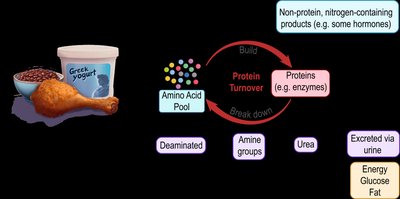
Protein Turnover and Amino Acid Pool
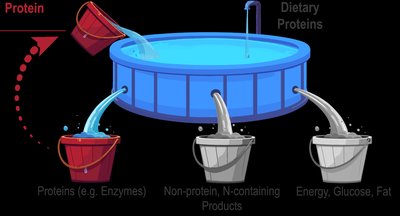
Protein turnover is the continuous process of breaking down and rebuilding proteins, allowing the body to adapt to changing needs. The amino acid pool consists of all available amino acids in the body, sourced from diet and protein turnover. Amino acids have three primary fates: building new proteins, synthesizing non-protein nitrogen-containing compounds, or being deaminated for energy, glucose, or fat production.
Dietary Guidelines for Proteins
Protein requirements vary by age, activity level, pregnancy, and dietary patterns. The Recommended Dietary Allowance (RDA) for adults is 0.8 g of protein per kilogram of body weight per day. The Acceptable Macronutrient Distribution Range (AMDR) is 10–35% of total daily calories from protein.

RDA:
AMDR: 10–35% of total daily calories from protein
Nitrogen balance is used to assess protein requirements. It is the state where nitrogen intake equals nitrogen excretion. Positive nitrogen balance occurs during growth, pregnancy, or recovery from illness; negative balance occurs during illness, injury, or inadequate protein intake.



Essential, Nonessential, and Conditionally Essential Amino Acids
Of the 20 amino acids, 9 are essential (must be obtained from the diet), 11 are nonessential (synthesized by the body), and some are conditionally essential under specific circumstances (e.g., illness).
Essential Amino Acids | Nonessential Amino Acids |
|---|---|
Histidine, Isoleucine, Leucine, Lysine, Methionine, Phenylalanine, Threonine, Tryptophan, Valine | Alanine, Arginine, Asparagine, Aspartic acid, Cysteine, Glutamic acid, Glutamine, Glycine, Proline, Serine, Tyrosine |
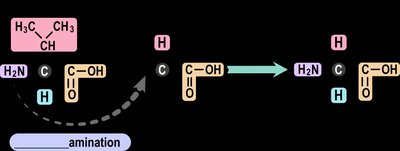
Transamination: The process by which the body produces nonessential amino acids by transferring an amino group from one molecule to another.
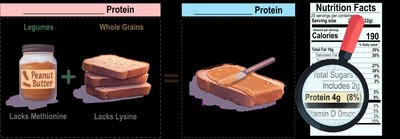
Protein Quality and Sources
Protein quality is determined by completeness (presence of all 9 essential amino acids) and digestibility. Complete proteins (mostly animal sources) contain all essential amino acids, while incomplete proteins (mostly plant sources) lack one or more. Mutual supplementation combines different incomplete proteins to form a complete protein.

High-Quality Proteins: Animal products, soy, quinoa
Low-Quality Proteins: Most plant sources (except soy and quinoa)
PDCAAS: Protein Digestibility Corrected Amino Acid Score ranks protein quality (max 100%).
Vegetarian and Vegan Diets
Vegetarianism excludes some or all animal products, while veganism excludes all animal-derived foods. These diets can be healthful but may require careful planning to avoid deficiencies in protein, vitamin B12, vitamin D, calcium, iron, and zinc.
Nutrient | Function | Vegetarian/Vegan Alternatives |
|---|---|---|
Protein | Growth, repair, structure | Soy, nuts, quinoa, beans, whole grains |
Vitamin B12 | DNA synthesis, nerve protection | Fortified foods, yeast, supplements |
Vitamin D & Calcium | Bone health | Fortified foods, sunlight |
Iron | Oxygen transport | Whole grains, dried fruit, beans, nuts, seeds, leafy greens |
Zinc | DNA/RNA synthesis, immunity | Whole grains, beans, nuts, seeds |

Protein-Related Disorders
Both protein deficiency and excess can have health consequences. Protein-energy malnutrition (PEM) includes marasmus (severe wasting) and kwashiorkor (edema due to protein deficiency). Excess protein intake may have conflicting health effects, with some research suggesting increased risk for certain diseases and others showing benefits.
Disorder | Details |
|---|---|
Marasmus | Chronic calorie deficiency, severe weight loss, low body mass |
Kwashiorkor | Adequate calories but protein deficiency, edema, tissue wasting |
Marasmic kwashiorkor | Combination of both, bloated limbs, and severe wasting elsewhere |
Note: Consuming too much or too little protein often means imbalanced intake of other nutrients.
