Textbook Question
Give an acceptable name for each amine.
(c)
1095
views

 Verified step by step guidance
Verified step by step guidance

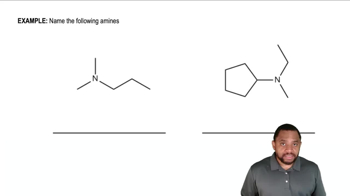

Give an acceptable name for each amine.
(c)
Predict the product of the following reactions.
(c)
We usually calculate Keq for acid–base reactions using pKa values.
(a) Derive an equation to calculate Keq using pKb values, then
(b) use it to calculate the equilibrium constant for the following reaction.
Which would you expect to be a stronger nucleophile, ethyl amine or diethyl amine? Why?
Label the amines shown as a 1° amine, 2° amine, 3° amine, or 4° ammonium ion.
a.
b.
c.
d.
Given the following names, draw the structure of the molecule.
c. (S)-2-methyloctan-4-amine