The 1H NMR spectra of three isomers with molecular formula C7H14O are shown here. Which isomer produces which spectrum?
b. <IMAGE>

 Verified step by step guidance
Verified step by step guidance
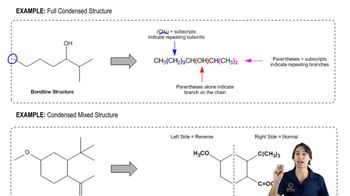
The 1H NMR spectra of three isomers with molecular formula C7H14O are shown here. Which isomer produces which spectrum?
b. <IMAGE>
An alkyl halide reacts with an alkoxide ion to form a compound whose 1H NMR spectrum is shown here. Identify the alkyl halide and the alkoxide ion. (Hint: See Section 9.15.)
<IMAGE>
Sketch the following spectra that would be obtained for 2-chloroethanol:
a. The 1H NMR spectrum for an anhydrous sample of the alcohol.
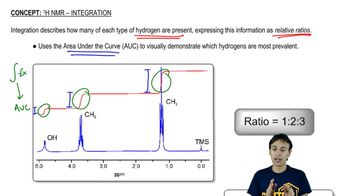
The 1H NMR spectrum of 2-propen-1-ol is shown here. Indicate the protons in the molecule that are responsible for each of the signals in the spectrum.
<IMAGE>
Draw a splitting diagram for the Hb proton if Jbc = 10 and Jba = 5.
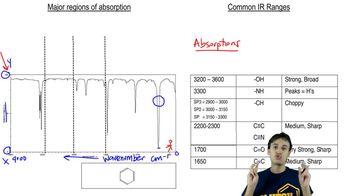
Identify each of the following compounds from its molecular formula and its IR and 1H NMR spectra:
b. C6H12O2
<IMAGE>