(i) Classify the following molecules as aromatic, nonaromatic, or antiaromatic.
(ii) For aromatic molecules, solve for n in Hückel’s rule. For all other molecules, explain which rule of aromaticity is being broken.
(d)

 Mullins 1st Edition
Mullins 1st Edition Ch. 23 - Benzene I: Aromatic Stability and Substitution Reactions
Ch. 23 - Benzene I: Aromatic Stability and Substitution Reactions Problem 61e
Problem 61e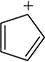
 Verified step by step guidance
Verified step by step guidance



(i) Classify the following molecules as aromatic, nonaromatic, or antiaromatic.
(ii) For aromatic molecules, solve for n in Hückel’s rule. For all other molecules, explain which rule of aromaticity is being broken.
(d)
(i) Classify the following molecules as aromatic, nonaromatic, or antiaromatic.
(ii) For aromatic molecules, solve for n in Hückel’s rule. For all other molecules, explain which rule of aromaticity is being broken.
(h)
(i) Classify the following molecules as aromatic, nonaromatic, or antiaromatic.
(ii) For aromatic molecules, solve for n in Hückel’s rule. For all other molecules, explain which rule of aromaticity is being broken.
(g)
(i) Classify the following molecules as aromatic, nonaromatic, or antiaromatic.
(ii) For aromatic molecules, solve for n in Hückel’s rule. For all other molecules, explain which rule of aromaticity is being broken.
(c)
(i) Classify the following molecules as aromatic, nonaromatic, or antiaromatic.
(ii) For aromatic molecules, solve for n in Hückel’s rule. For all other molecules, explain which rule of aromaticity is being broken.
(b)
(i) Classify the following molecules as aromatic, nonaromatic, or antiaromatic.
(ii) For aromatic molecules, solve for n in Hückel’s rule. For all other molecules, explain which rule of aromaticity is being broken.
(f)