Textbook Question
Which nucleophile would be more reactive in the solvent given?
(a)
966
views

 Mullins 1st Edition
Mullins 1st Edition Ch. 12 - Substitution and Elimination: Reactions of Haloalkanes
Ch. 12 - Substitution and Elimination: Reactions of Haloalkanes Problem 21c
Problem 21c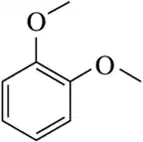
 Verified step by step guidance
Verified step by step guidance

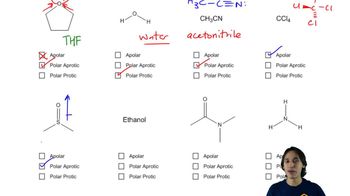

Which nucleophile would be more reactive in the solvent given?
(a)
Identify the following solvents as polar protic, polar aprotic, or nonpolar.
(a)
Would the following nucleophiles be more likely to participate in an SN1 or SN2 reaction?
(e) H2O
Would the following nucleophiles be more likely to participate in an SN1 or SN2 reaction?
(f)
Which nucleophile would be more reactive in the solvent given?
(b)
Identify the following solvents as polar protic, polar aprotic, or nonpolar.
(e)