Convert the perspective formula to a skeletal structure.

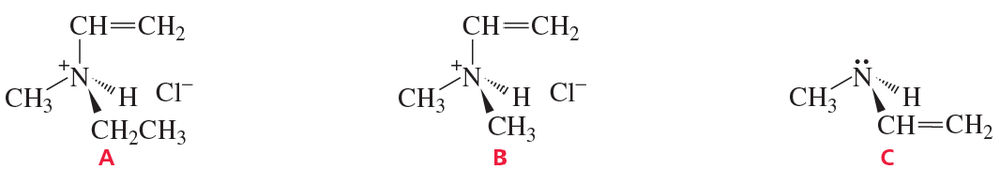
Explain why compound A has two stereoisomers but compounds B and C exist as single compounds.
 Verified step by step guidance
Verified step by step guidance
Verified video answer for a similar problem:
Key Concepts
Stereoisomerism

Chirality

Geometric Isomerism
Disregarding stereoisomers, draw the structures of all alkenes with molecular formula C5H10. Which ones can exist as cis–trans isomers?
Limonene exists as two different stereoisomers. The R enantiomer is found in oranges and lemons, and the S enantiomer is found in spruce trees. Which of the following is found in oranges and lemons?
Convert the Fischer projection to a perspective formula.
Draw all possible stereoisomers for each of the following. Indicate those compounds for which no stereoisomers are possible.
a. 1-bromo-2-chlorocyclohexane
b. 2-bromo-4-methylpentane
Threonine, an amino acid, has four stereoisomers. The stereoisomer found in nature is (2S,3R)-threonine. Which of the following structures represents the naturally occurring amino acid?
