Textbook Question
Use oxidation numbers to determine which of the following are redox reactions.
(d)
1025
views


 Verified step by step guidance
Verified step by step guidance

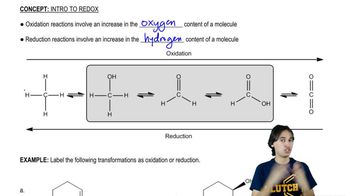

Use oxidation numbers to determine which of the following are redox reactions.
(d)
Determine the oxidation number of each carbon indicated by an arrow.
(h)
Determine the oxidation number of each carbon indicated by an arrow.
(f)
Use oxidation numbers to determine which of the following are redox reactions.
(c)
Use oxidation numbers to determine which of the following are redox reactions.
(e)
Determine the oxidation number of each carbon indicated by an arrow.
(g)