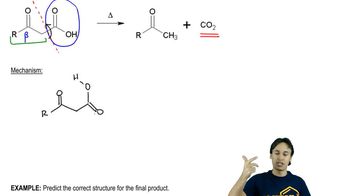
Textbook Question
Explain why [1,3] sigmatropic migrations of hydrogen cannot occur under thermal conditions, but [1,3] sigmatropic migrations of carbon can.
1186
views

 Verified step by step guidance
Verified step by step guidance

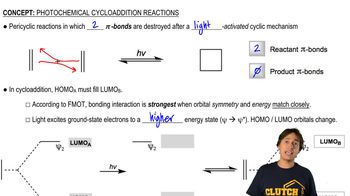
Explain why [1,3] sigmatropic migrations of hydrogen cannot occur under thermal conditions, but [1,3] sigmatropic migrations of carbon can.
b. Will thermal 1,5-migrations of carbon occur with retention or inversion of configuration?
Explain why the hydrogen and the methyl substituent are trans to one another after photochemical ring closure of provitamin D3 to form 7-dehydrocholesterol.
Show how 5-methyl-1,3-cyclopentadiene rearranges to form 1-methyl-1,3-cyclopentadiene and 2-methyl-1,3-cyclopentadiene.
Does the [1,7] sigmatropic rearrangement that converts provitamin D3 to vitamin D3 involve suprafacial or antarafacial rearrangement?
Account for the difference in the products obtained under photochemical and thermal conditions: