Textbook Question
Propose a mechanism for the following reaction:
767
views

![Chemical structure showing a [1,3] sigmatropic rearrangement with products labeled A and B.](https://static.studychannel.pearsonprd.tech/courses/organic-chemistry/thumbnails/7d32594d-bf7d-4007-b4a3-ad32290b6c43)
 Verified step by step guidance
Verified step by step guidance
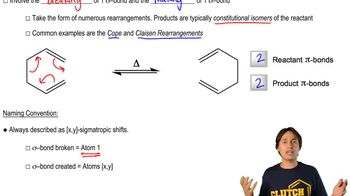


Propose a mechanism for the following reaction:
Draw the product of each of the following reactions:
e.
If the compounds shown here are heated, one will form one product from a [1,3] sigmatropic rearrangement and the other will form two products from two different [1,3] sigmatropic rearrangements. Draw the products of the reactions.
Draw the product of each of the following reactions:
d.
Draw the product formed when each of the following compounds undergoes an electrocyclic reaction
b. under photochemical conditions.
1.
2.
Draw the product of each of the following sigmatropic rearrangements:
c.