At what pH is the concentration of a compound, with a pKa = 8.4, 100 times greater in its acidic form than in its basic form? At what pH is 50% of a compound, with a pKa = 7.3, in its basic form?

 Bruice 8th Edition
Bruice 8th Edition Ch. 2 - Acids and Bases: Central to Understanding Organic Chemistry
Ch. 2 - Acids and Bases: Central to Understanding Organic Chemistry Problem 43d
Problem 43dA naturally occurring amino acid such as alanine has a group that is a carboxylic acid and a group that is a protonated amine. The pKa values of the two groups are shown.
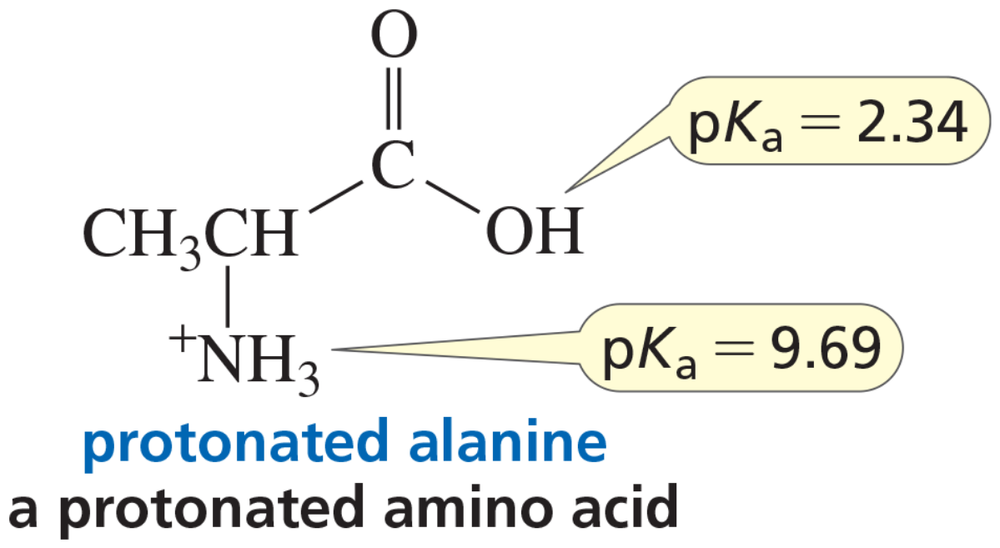
d. Draw the structure of alanine in a solution at pH = 12.
 Verified step by step guidance
Verified step by step guidance
Verified video answer for a similar problem:
Key Concepts
Amino Acids

pKa and Ionization

Zwitterion Form
A naturally occurring amino acid such as alanine has a group that is a carboxylic acid and a group that is a protonated amine. The pKa values of the two groups are shown.
e. Is there a pH at which alanine is uncharged (that is, neither group has a charge)?
f. At what pH does alanine have no net charge (that is, the amount of negative charge is the same as the amount of positive charge)?
A naturally occurring amino acid such as alanine has a group that is a carboxylic acid and a group that is a protonated amine. The pKa values of the two groups are shown.
c. Draw the structure of alanine in a solution at physiological pH (pH 7.4).
Indicate whether an alcohol (ROH) with a pKa value of 15 has more charged or more neutral molecules in a solution with the pH values given in Problem 41.
3. pH = 7
4. pH = 10
5. pH = 13
Indicate whether an alcohol (ROH) with a pKa value of 15 has more charged or more neutral molecules in a solution with the pH values given in Problem 41.
1. pH = 1
2. pH = 3
3. pH = 5
At what pH is the concentration of a compound, with a pKa = 4.6, 10 times greater in its basic form than in its acidic form?