The following 1H NMR spectra are for four compounds, each with molecular formula of C6H12O2. Identify the compounds.
c. <IMAGE>

 Verified step by step guidance
Verified step by step guidance

The following 1H NMR spectra are for four compounds, each with molecular formula of C6H12O2. Identify the compounds.
c. <IMAGE>
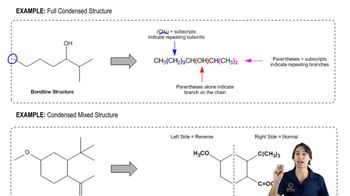
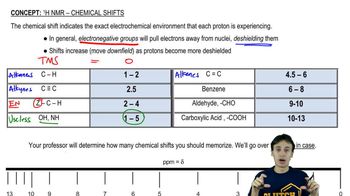
How many signals are produced by each of the following compounds in its
a. 1H NMR spectrum?
4.
5.
6.
When compound A (C5H12O) is treated with HBr, it forms compound B (C5H11Br). The 1H NMR spectrum of compound A has a 1H singlet, a 3H doublet, a 6H doublet, and two 1H multiplets. The 1H NMR spectrum of compound B has a 6H singlet, a 3H triplet, and a 2H quartet. Identify compounds A and B.
The following 1H NMR spectra are for four compounds, each with molecular formula of C6H12O2. Identify the compounds.
d. <IMAGE>
Identify each of the following compounds from its molecular formula and its IR and 1H NMR spectra:
b.C6H14O
<IMAGE>
How many signals are produced by each of the following compounds in its
b. 13C NMR spectrum?
6.