Draw the products of each of the following SN2/E2 reactions. If the products can exist as stereoisomers, show which stereoisomers are formed.
c. (3S,4R)-3-bromo-4-methylhexane + CH3O−

 Bruice 8th Edition
Bruice 8th Edition Ch. 9 - Substitution and Elimination Reactions of Alkyl Halides
Ch. 9 - Substitution and Elimination Reactions of Alkyl Halides Problem 114
Problem 114 Verified step by step guidance
Verified step by step guidance

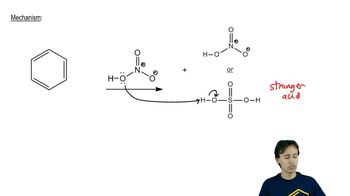

Draw the products of each of the following SN2/E2 reactions. If the products can exist as stereoisomers, show which stereoisomers are formed.
c. (3S,4R)-3-bromo-4-methylhexane + CH3O−
Draw the elimination products for each of the following E2 reactions; if the products can exist as stereoisomers, indicate which stereoisomers are obtained.
d. (2R,3R)-2-chloro-3-methylpentane + high concentration of CH3O-
Draw the major elimination product that would be obtained from each of the following reactants with a strong base and with a weak base:
a.
Draw the products of each of the following SN2/E2 reactions. If the products can exist as stereoisomers, show which stereoisomers are formed.
a. (3S,4S)-3-bromo-4-methylhexane + CH3O−
Draw the products of each of the following SN2/E2 reactions. If the products can exist as stereoisomers, show which stereoisomers are formed.
b. (3R,4R)-3-bromo-4-methylhexane + CH3O−
Draw the elimination products for each of the following E2 reactions; if the products can exist as stereoisomers, indicate which stereoisomers are obtained.
e. 3-chloro-3-ethyl-2,2-dimethylpentane + high concentration of CH3CH2O-