Which reaction in each of the following pairs takes place more rapidly? (EtOH is ethyl alcohol; Et2O is diethyl ether.)
d.
e.

 Bruice 8th Edition
Bruice 8th Edition Ch. 9 - Substitution and Elimination Reactions of Alkyl Halides
Ch. 9 - Substitution and Elimination Reactions of Alkyl Halides Problem 56a,b,c
Problem 56a,b,c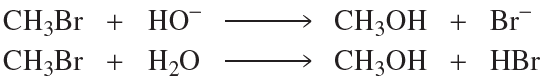
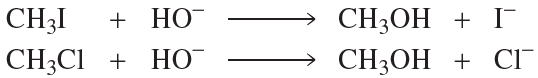
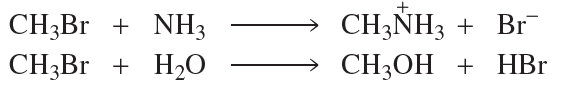
 Verified step by step guidance
Verified step by step guidance



Which reaction in each of the following pairs takes place more rapidly? (EtOH is ethyl alcohol; Et2O is diethyl ether.)
d.
e.
Would you expect acetate ion (CH3CO2−) to be a better nucleophile in an SN2 reaction with an alkyl halide carried out in methanol or in dimethyl sulfoxide?
Under which of the following reaction conditions will (R)-1-chloro-1-phenylethane form the most (R)-1-phenyl-1-ethanol: in water or in 1.0 M HO−?
How will the rate of each of the following SN2 reactions change if it is carried out in a more polar solvent?
b.
How will the rate of each of the following SN2 reactions change if it is carried out in a more polar solvent?
a.
How will the rate of each of the following SN2 reactions change if it is carried out in a more polar solvent?
c.