Textbook Question
Draw the product of each of the following reactions:
2.
982
views

 Bruice 8th Edition
Bruice 8th Edition Ch. 9 - Substitution and Elimination Reactions of Alkyl Halides
Ch. 9 - Substitution and Elimination Reactions of Alkyl Halides Problem 120c
Problem 120c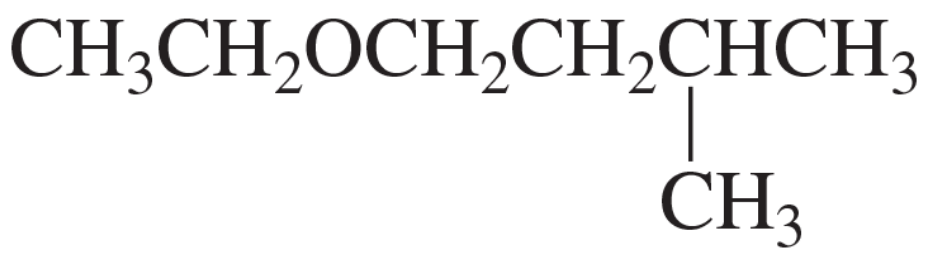
 Verified step by step guidance
Verified step by step guidance



Draw the product of each of the following reactions:
2.
The reaction of an alkyl chloride with potassium iodide is generally carried out in acetone to maximize the amount of alkyl iodide that is formed. Why does the solvent increase the yield of alkyl iodide? (Hint: Potassium iodide is soluble in acetone, but potassium chloride is not.)
Draw the product of each of the following reactions:
1.
Propose a mechanism for the following reaction:
Give two sets of reactants (each set including an alkyl halide and a nucleophile) that could be used to synthesize the following alkyne:
CH3CH2C≡CCH2CH2CH2CH3
Explain why tetrahydrofuran can solvate a positively charged species better than diethyl ether can.