Textbook Question
Predict the products formed when cyclohexanecarbaldehyde reacts with the following reagents.
(a) PhMgBr, then H3O+
1428
views

 Verified step by step guidance
Verified step by step guidance
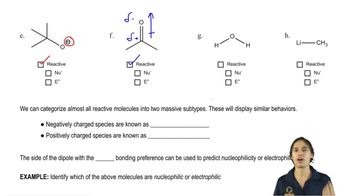

Predict the products formed when cyclohexanecarbaldehyde reacts with the following reagents.
(a) PhMgBr, then H3O+
Predict the products formed when cyclohexanone reacts with the following reagents.
(l) acidic hydrolysis of the product from (k)
Predict the products formed when cyclohexanone reacts with the following reagents.
(h) sodium acetylide, then mild H3O+
Predict the products formed when cyclohexanone reacts with the following reagents.
(k) sodium cyanide
Predict the products formed when cyclohexanone reacts with the following reagents.
(f) PhMgBr and then mild H3O+
Predict the products formed when cyclohexanone reacts with the following reagents.
g. Tollens reagent