Textbook Question
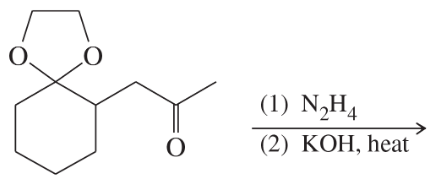
Predict the major products of the following reactions:
(d)
799
views

 Verified step by step guidance
Verified step by step guidance



Predict the major products of the following reactions:
(d)
Predict the major products of the following reactions:
(b)
Draw structures of the following derivatives.
(a) the 2,4-dinitrophenylhydrazone of benzaldehyde
Propose a mechanism for both parts of the Wolff–Kishner reduction of cyclohexanone: the formation of the hydrazone, and then the base-catalyzed reduction with evolution of nitrogen gas.
Predict the major products of the following reactions:
(a)
Draw structures of the following derivatives.
(b) the semicarbazone of cyclobutanone